PROTEIN TRANSDUCTION DOMAINS (PTDs)
Cell-penetrating peptides (CPPs) constitute a promising tool for the cellular import of drug cargos. They have been successfully applied for in vitro and in vivo delivery of a variety of therapeutic molecules including plasmids, DNA, oligonucleotides, siRNA, PNA, proteins, peptides, low molecular weight drugs, liposomes, and nanoparticles.
Introduction
Delivery of therapeutic agents into cells through their membrane (cellular uptake) is a topic of profound importance to medicinal chemists and for the pharmaceutical industry and has always proved to be a challenging task especially in the delivery of large molecules. The plasma membrane prevents direct translocation of hydrophilic macromolecules by acting as a barrier to efficient and controlled intracellular delivery. A drug must be either highly lipophilic or very small to stand a chance of cellular internalization and it is difficult to ascertain a generic mechanism for drug uptake. These restrictions mean that the repertoire of possible drug molecules is limited. Similarly, novel therapeutic approaches such as gene and protein therapy also have limited potential due to the cell-impermeable nature of peptides and oligonucleotides. The existing methods for delivery of macromolecules, such as viral vectors and membrane perturbation techniques, can result in high toxicity, immunogenicity and low delivery yield.
Discovery and potential of cell-permeable peptides
In 1988, Frankel and Pabo observed the remarkable ability of HIV-Tat protein to enter cells and translocate into the nucleus. Mutagenesis studies of the protein showed that the region between residues 47–57 (sequence: YGRKKRRQRRR) is important for cellular uptake. Soon after, in 1991, the group of Prochiantz and Derossi demonstrated that the antennapedia homeodomain protein of Drosophila melanogaster could be internalized by neuronal cells. This discovery subsequently led to the identification of a hexadecapeptide, penetratin, derived from the third helix of the homeodomain of antennapedia. Since then, the number of known natural and synthetic peptides with cell-penetrating capabilities has continued to grow (Table 1).
These peptides which are able to penetrate the cell membrane and enter the cell are known as cell-penetrating peptides. They are also known as protein transduction domains (PTDs), membrane translocating sequences (MTSs) or Trojan peptides. PTDs were identified in transcription factors, bacterial or viral surface proteins, toxins, amphipathic helix-forming peptides and in ligands of membrane-bound receptors or adhesion proteins. CPPs can be broadly classified as proteinderived, chimeric (synthetic peptides combining partial sequneces of natural peptides), or synthetic, as shown in Table 1.
These short peptides with less than 40 amino acids share common features such as positively charged amino acids, hydrophobicity and amphipathicity. The discovery and the ability of these peptides to traverse the cell membrane opened up a new avenue for drug delivery. Transport of therapeutically significant biomolecules across the membrane into the cell can be facilitated by attaching them to CPPs. A major breakthrough in the CPP field came from the first proofs-of-concept of their in vivo application, by the groups of Dowdy, for the delivery of small peptides and large proteins and of Langel, for delivery of peptide nucleic acids (PNAs) using the chimeric peptide transportan, derived from the N-terminal fragment of the neuropeptide galanin, linked to mastoparan, a wasp venom peptide. Since then, several CPPs that are able to trigger the movement of a cargo across the cell membrane into the cytoplasm have been designed.
Numerous cargo molecules have been attached to CPPs for cellular delivery. These include plasmid, DNA, oligonucleotides, siRNA, PNAs, proteins, peptides, liposomes, low molecular weight drugs, antibodies, nanoparticles, antibiotics, enzymes and enzyme substrates.
| Protein-derived | Chimeric | De novo designed |
|---|---|---|
| HIV-tat Protein | Galanin-Mastoparan | Oligoarginines |
| HIV-1 tat (47-57) | Transportan | Octa/nona-arginine |
| YGRKKRRQRRR | GWTLNSAGYLLGKINLKALAALAKKIL | RRRRRRRR(R) |
| Antennapedia homeoprotein | HIV rev -SV40 large T-antigen | MAP |
| Penetratin | Pep-1 | KLAL |
| (Antp homeobox (43-58) amide) | KETWWETWWTEWSQPKKKRKV | KLALKLALKALKAALKLA |
| RQIKIWFQNRRMKWKK-NH? | ||
| Herpes simplex Virus HSV-1 | HIV gp41-SV40 T-antigen | KADY |
| VP-22 | MPG | |
| DAATATRGRSAASRPTERPRAPARSASRPRRPVD | GALFLGFLGAAGSTMGAWSQPKKKRKV | Ac-GLWRALWRLLRSLWRLLWKAcysteamide |
| VE-Cadherin | p14 ARF | KALA |
| pVEC | M-918 | |
| LLIILRRRIRKQAHAHSK-NH2 | MVTVLFRRLRIRRASGPPRVRV-NH2 | WEAKLAKALAKALAKHLAKALAKALKACEA |
Table 1: Examples of different CPPs that are protein-derived, chimeric sequences, or de novo designed peptides.
Methods of attaching CPPs to cargos
CPPs are usually connected via a covalent linkage to the cargo molecule. Proteins and peptides can be attached to CPPs through a disulfide bond (by modifying CPP and peptide/protein with cysteine) or through cross-linkers. Most CPP-nucleic acid complexes that have been proposed so far are formed through covalent bonding. Different strategies include cleavable disulfide, amide, thiazolidine, oxime and hydrazine linkages. Short interfering RNA (siRNA) can be covalently linked to transportan and penetratin by disulfide linkage at the 5’-end of the sense strands of siRNA to target luciferase or eGFP mRNA reporters.
A stable covalent linkage between the cargo and CPP is not always necessary for translocation as simple mixing of two entities was shown to be efficient. The synthetic covalent bond between CPP and nucleic acid may alter the biological activity of the latter. In 1997, the first non-covalent CPP for delivery of nucleic acids, called MPG (see Table 1) was designed by the group of Heitz and Divita closely followed by development of Pep-1 for non-covalent cellular delivery of proteins and peptides by Morris et al. in 2001. The groups of Wender and of Futaki demonstrated that oligoarginine sequences (Arg8) were sufficient to drive molecules into cells and proposed that their uptake mechanism involves a bidentate hydrogenbonding interaction between the guanidinium moieties of the arginine residues and phosphate groups in the membrane.
Thus, a new non-covalent strategy requiring no chemical modification with short amphipathic CPPs, like MPG and Pep-1 as carriers has been successfully applied for delivery of cargoes. These non-covalent conjugates are formed through either electrostatic or hydrophobic interactions. With this method, cargoes such as nucleic acids and proteins could be efficiently delivered while maintaining full biological activity. MPG forms highly stable complexes with siRNA with a low degradation rate and can be easily functionalized for specific targeting, which are major advantages compared with the covalent CPP technology.
Crotamine, another natural CPP has been isolated from the venom of the South American rattlesnake Crotalus durissus terrificus.
Mechanism of CPP translocation across the cell membrane
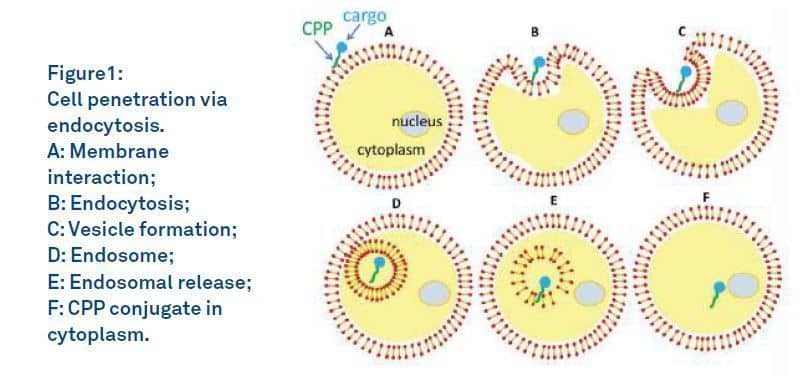
The exact molecular pathways underlying cellular uptake of a cargo attached to a CPP are not clear. Different CPPs have varying hydrophobicity, charge and amphiphilicity. The size and chemical properties of cargos are also different. Hence, generalizing the interaction of these complex molecules and cell membrane is not easy. For docking and cellular uptake, two major mechanisms have been considered: the endosomal pathways composed of endocytotic entry followed by endosomal escape, and direct cell membrane penetration. Peptides that have a high affinity for membranes have a higher propensity to be internalized by a non-endocytic mechanism than peptides with a lower affinity. CPPs with low molecular weight cargos may also enter without vesicle formation and facilitate access to all intracellular compartments. Different stages of cell penetration via endocytosis are depicted in Figure 1. According to this mechanism, CPPs are first simply adsorbed at the cell, followed by endocytosis of membrane, vesicle formation, formation of endosomes in which the conjugate is trapped, and endosomal release.
Drug delivery: CPP-drug conjugates in clinical trials
Research and clinical studies on the transport and delivery of therapeutics into cellular targets using cell-penetrating peptides has been progressing well in recent years. Several companies started working on clinical development of CPPs, for topical and systemic administration of different therapeutic molecules. The first CPP clinical trial was initiated by Cellgate Inc. for topical delivery of cyclosporine linked to polyarginine (CGC1072) and entered phase II trials in 2003 for the treatment of psoriasis. This is an example of local application of a CPP-drug conjugate (local CPP-mediated delivery). However, despite an efficient uptake of the chimera, the release of the free drug was not rapid enough to compete with clearance. A list of different CPP-based drugs which entered clinical trial is shown in Table 2.
The therapeutic 28-amino acid cell-penetrating peptide p28 is derived from azurin, a redox protein secreted from the pathogen Pseudomonas aeruginosa, produces a posttranslational increase in p53 by inhibiting its ubiquitination in cancerous cells. In few of these cases therapeutic agents are covalently linked either directly or through a linker to the CPP carrier. In KAI-9803, KAI-1678 and KAI-1455, the cargo peptide is attached to Tat peptide via a disulfide bond between additional cysteines at the N-termini of both entities. The cargo peptides SFNSYELGSL and EAVSLKPTC are δ protein kinase C (δPKC) and ε protein kinase C (εPKC) specific inhibitors, respectively and HDAPIGYD is a εPKC activator peptide. DTS-108 is a Vectocell® peptide-SN38 prodrug generated by esterification of the 10-hydroxyl group of SN38 to a heterobifunctional cross-linker (BCH) linked to Vectocell® peptide DPV1047 (CVKRGLKLRHVRPRVTRMDV).
| CPP | Sequence | Drug | Structure / Sequence | Indication / Status |
|---|---|---|---|---|
| p28 Azurin (50-77) | LSTAADMQGVVTDGMASGLDKDYLKPDD | Azurin-p28 (NSC745104) | LSTAADMQGVVTDGMASGLDKDYLKPDD | Progressive CNS tumors/ phase 1 |
| TAT 48-57 | GRKKRRQRRR | XG-102 (D-JNKI-1) | GRKKRRQRRRPP- RPKRPTTLNLFPQVPRSQDT | Hearing loss/stroke |
| TAT 4757 | YGRKKRRQRRR | KAI-9803 (Delcasertib) | CYGRKKRRQRRR/ CSFNSYELGSL | Myocardial infarction/ phase 2b |
| TAT 4757 | YGRKKRRQRRR | KAI-1678 | CYGRKKRRQRRR/ CEAVSLKPT | Pain/phase 2a |
| TAT 47-57 | YGRKKRRQRRR | KAI-1455 | CYGRKKRRQRRR/ CHDAPIGYD | Cytoprotection/phase 1 |
| Vectocell® peptide | CVKRGLKLRHVRPRVTRMDV | DTS-108 | SN38-BCHCVKRGLKLRHVRP | Colon cancer/phase 1 |
Conclusions
Extensive research on the design and in vivo applications of cell-penetrating peptides during the last two decades has demonstrated the potential of these compounds in molecular medicine. With the aid of CPPs, delivery of therapeutic biomolecules such as oligonucleotides and proteins can be better managed in terms of efficiency, cytotoxicity and biocompatibility. CPPs can associate with an extensive range of cargo types either via covalent linkage or noncovalent interaction. One of the limitations of CPPs is the non-specific cellular uptake which limits their potential for drug delivery to specific cellular targets such as tumor cells. Fortunately, a number of recently developed CPPs have shown high affinity for specific cell types or intracellular destinations. A recently discovered CPP known as ‘crotamine’ has shown unusually high affinity for actively proliferating cells. Another example lies with MPG, a synthetic CPP derived from the SV40 virus. Originally designed for nuclear delivery of siRNA, it has recently been altered to target the cytoplasm. Recently, “activatable” CPPs were introduced to address the problem of tissue non-specificity of CPPs. Constructs have been described that are “activated” either outside or inside the cells. Another solution to the problem of cellular non-specificity of CPPs will be, starting from the sequences of well-established cell-targeting peptides, to design new peptides that combine both targeting and internalization properties.
Subscribe to our newsletter
"*" indicates required fields
References
A.D. Frankel and C.O. Pabo
Cellular uptake of the Tat protein from human immunodeficiency virus.
Cell 55, 1189–1193 (1988)
M. Green and P.M. Loewenstein
Autonomous functional domains of chemically synthesized human immunodeficiency virus tat transactivator protein.
Cell 55, 1179–1188 (1988)
D. Derossi et al.
The third helix of the Antennapedia homeodomain translocates through biological membranes.
J. Biol. Chem. 269, 10444-10450 (1994)
E. Vives et al.
A truncated HIV-1 Tat protein basic domain rapidly translocates through the plasma membrane and accumulates in the cell nucleus.
J. Biol. Chem. 272, 16010–16017 (1997)
M. Pooga et al.
Galanin-based peptides, galparan and transportan, with receptordependent and independent activities.
Ann. N. Y. Acad. Sci. 863, 450-453 (1998)
L. Chen et al.
Opposing cardioprotective actions and parallel hypertrophic effects of delta PKC and epsilon PKC.
Proc. Natl. Acad. Sci. U. S. A. 98, 11114-11119 (2001)
S. Futaki et al.
Arginine-rich peptides. An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery.
J. Biol. Chem. 276, 5836-5840 (2001)
M.C. Morris et al.
A peptide carrier for the delivery of biologically active proteins into mammalian cells.
Nat. Biotechnol. 19, 1173-1176 (2001)
S. Deshayes et al.
Insight into the mechanism of internalization of the cell-penetrating carrier peptide Pep-1 through conformational analysis.
Biochemistry 43, 1449-1457 (2004)
C. De Coupade et al.
Novel human-derived cell-penetrating peptides for specific subcellular delivery of therapeutic biomolecules.
Biochem. J. 390, 407-418 (2005)
S. Deshayes et al.
Cell-penetrating peptides: tools for intracellular delivery of therapeutics.
Cell. Mol. Life Sci. 62,1839–1849 (2005)
S. El-Andaloussi et al.
Cell-penetrating peptides: mechanism and applications.
Curr. Pharm. Design 11, 3597–3611 (2005)
F. Simeoni et al.
Peptide-based strategy for siRNA delivery into mammalian cells.
Methods Mol. Biol. 309, 251–264 (2005)
M. Zorko and Ü. Langel
Cell-penetrating peptides: mechanism and kinetics of cargo delivery.
Adv. Drug Deliv. Rev. 57, 529-545 (2005)
A. Elmquist et al.
Structure-activity relationship study of the cell-penetrating peptide pVEC.
Biochim. Biophys. Acta 1758, 721- 729 (2006)
E.A. Goun et al.
Intracellular cargo delivery by an octaarginine transporter adapted to target prostate cancer cells through cell surface protease activation.
Bioconjug. Chem. 17, 787-796 (2006)
S. El-Andaloussi et al.
A novel cell-penetrating peptide, M918, for efficient delivery of proteins and peptide nucleic acids.
Mol. Ther. 15, 1820-1826 (2007)
M.C. Morris et al.
Cell-penetrating peptides: from molecular mechanisms to therapeutics.
Biol. Cell. 100, 201–217 (2008)
L. Crombez et al.
A new potent secondary amphipathic cell-penetrating peptide for siRNA delivery into mammalian cells.
Mol. Ther. 17, 95-103 (2009)
A. Eguchi and S.F. Dowdy
siRNA delivery using peptide transduction domains.
Trends Pharmacol. Sci. 30, 341-345 (2009)
F. Heitz et al.
Twenty years of cell-penetrating peptides: from molecular mechanisms to therapeutics.
Br. J. Pharmacol. 157, 195–206 (2009)
L.E. Yandek et al.
Wasp mastoparans follow the same mechanism as the cell-penetrating peptide transportan 10.
Biochemistry 48, 7342-7351 (2009)
E. Dupont et al.
Penetratin story: an overview.
Methods Mol. Biol. 683, 21-29 (2011)
K. Splith and I. Neundorf
Antimicrobial peptides with cellpenetrating peptide properties and vice versa.
Eur. Biophys. J. 40, 387-397 (2011)
F.D. Nascimento et al.
The natural cell-penetrating peptide crotamine targets tumor tissue in vivo and triggers a lethal calciumdependent pathway in cultured cells.
Mol. Pharm. 9, 211-221 (2012)
C. Bechara and S. Sagan
Cell-penetrating peptides: 20 years later, where do we stand?
FEBS Lett. 587, 1693-1702 (2013)
J.E. Moodie et al.
A single-center, randomized, doubleblind, active, and placebo-controlled study of KAI-1678, a novel PKCepsilon inhibitor, in the treatment of acute postoperative orthopedic pain.
Pain Med. 14, 916-924 (2013)
T. Yamada et al.
p28, a first in class peptide inhibitor of cop1 binding to p53.
Br. J. Cancer. 108, 2495-504 (2013)
Y.S. Choi and A.E. David
Cell penetrating peptides and the mechanisms for intracellular entry.
Curr. Pharm. Biotechnol. 15, 192-199 (2014)
J. Pae et al.
Translocation of cell-penetrating peptides across the plasma membrane is controlled by cholesterol and microenvironment created by membranous proteins.
J. Control. Release 192, 103-113 (2014)
S. Reissmann
Cell penetration: scope and limitations by the application of cell-penetrating peptides.
J. Pept. Sci. 20, 760-784 (2014)
D. Sun et al.
Effect of arginine-rich cell penetrating peptides on membrane pore formation and life-times: a molecular simulation study.
Phys. Chem. Chem. Phys. 16, 20785- 20795 (2014)
Z. Wang et al.
CPP-mediated protein delivery in a noncovalent form: proof-of-concept for percutaneous and intranasal delivery.
Protein Pept. Lett. 21, 1129-1136 (2014)
J.S. Bahnsen et al.
Cell-penetrating antimicrobial peptides – prospectives for targeting intracellular infections.
Pharm. Res. 32, 1546-1556 (2015)
Y.W. Huang et al.
Delivery of nucleic acids and nanomaterials by cell-penetrating peptides: opportunities and challenges.
Biomed. Res. Int. 2015, 834079 (2015)
K. Pärn et al.
The antimicrobial and antiviral applications of cell-penetrating peptides.
Methods Mol. Biol. 1324, 223-245 (2015)
M. Pooga and Ü. Langel
Classes of cell-penetrating peptides.
Methods Mol. Biol. 1324, 3-28 (2015)