Ribosomally synthesized antimicrobial peptides (AMPs) constitute a structurally diverse group of molecules found virtually in all organisms. Most antimicrobial peptides contain less than 100 amino acid residues, have a net positive charge, and are membrane active. They are major players in the innate immune defense but can also have roles in processes as chemokine induction, chemotaxis, inflammation, and wound healing. In addition to their antimicrobial effects, many of them show antiviral and antineoplastic activities.
Introduction
AMPs are a heterogeneous group of relatively small molecules usually containing less than a hundred amino acids. They were first described in the 1960’s by Zeya and Spitznagel in polymorphonuclear leukocyte lysosomes.
To date, more than 2600 AMPs have been identified and registered in databases. They are produced by nearly all groups of organisms, including bacteria, fungi, plants, and animals. Many vertebrate AMPs are secreted by epithelial surfaces such as the tracheal, lingual, or intestinal mucosa of mammals or the skin of amphibia. Some are expressed in neutrophils, monocytes, and macrophages.
AMPs are involved in both animal and plant immune defense systems. Constitutively expressed or induced they play a key role in the first line of defense against microbial intruders.
Structure/Classification
AMPs can be classified on the basis of their amino acid composition and structure. Two major groups of AMPs can be distinguished. The first group consists of linear molecules which either tend to adopt α-helical structure or are enriched in certain amino acids such as arginine, glycine, histidine, proline, and tryptophan. The second group consists of cysteine-containing peptides which can be divided into single or multiple disulfide structures. In many cases, the presence of disulfide bridges is required for antimicrobial activity.
Most AMPs are cationic peptides, but there are also anionic peptides such as dermcidin, an aspartic acid-rich peptide from human and maximin H5 from amphibian skin. Other non-cationic AMPs include fragments from neuropeptide precursor molecules such as proenkephalin A, aromatic dipeptides primarily isolated from dipteran larvae, or peptides derived from oxygen-binding proteins from arthropod or annelid species.
Mode of Action
Most AMPs act by provoking an increase in plasma membrane permeability. They preferentially target microbial versus mammalian cells. Selectivity is influenced by several factors such as differences in membrane composition: membranes of many bacterial pathogens contain negatively charged lipid moieties such as phosphatidylglycerol (PG), cardiolipin, and phosphatidylserine (PS), whereas mammalian membranes, commonly enriched in phosphatidylethanolamine (PE), phosphatidylcholine (PC) and sphingomyelin, are generally neutral in net charge.
The presence of sterols such as cholesterol and ergesterol within the membrane may be a further means by which AMPs can distinguish between mammalian or fungal cells and prokaryotes. A first step in the mechanism of membrane permeabilization is the electrostatic interaction between the positively charged AMP with the negatively charged membrane surface of the microorganism. Subsequent disruption of the membrane by creation of pores within the microbial membrane ultimately results in cell death of the organism due to leakage of ions, metabolites, cessation of membrane-coupled respiration, and biosynthesis.
Several models for pore formation such as the Barrel-Stave, the Toroidal or Wormhole Model, and the Carpet Model have been proposed (Fig. 1).
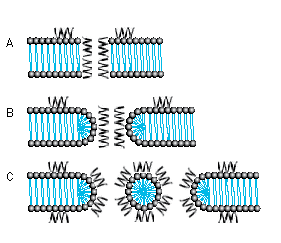
Fig. 1. Mode of Action A Barrel-Stave Model B Toroidal Pore or Wormhole Model C Carpet Model
The Barrel-Stave Model
The Barrel-Stave model describes a mechanism in which AMPs form a barrellike pore within the bacterial membrane with the individual AMPs or AMP complexes being the staves. Arranged in this manner, the hydrophobic regions of the AMPs point outwards towards the acyl chains of the membrane whereas the hydrophilic areas form the pore.
The Toroidal Pore or Wormhole Model
The pores described by this model differ from those of the Barrel-Stave model. Primarily, the outer and inner leaflet of the membrane are not intercalated in the transmembrane channel.
The Carpet Model
A different mechanism is proposed in the Carpet model where AMPs first cover the outer surface of the membrane and then disrupt the membrane like detergents by forming micelle-like units. Certain AMPs penetrate the bacterial membrane without channel formation. They act on intracellular targets by e.g. inhibiting nucleic acid and/or protein synthesis.
Resistance
Resistance to AMPs can either be constitutive or inducible. Inherited resistance mechanisms include altered surface charge, active efflux, production of peptidases or trapping proteins, and modification of host cellular processes. For instance, Staphylococcus aureus manages to reduce the overall cell surface charge by esterification of the cell wall component teichoic acid with D-alanine and thereby increases its resistance against human AMPs. Another example for changing the surface net charge is the production of cationic lysine-substituted phosphatidylglycerol (L-PG) found in certain Staphylococcus aureus strains. In Gram-negative bacteria, addition of 4-aminoarabinose (Ara4N) to the phosphate group of the lipid A backbone or increased acylation of lipopolysaccharides (LPS) are important mechanisms of AMP resistance. Exposure to AMPs may also induce stress responses by which microorganisms try to survive. Inducible regulatory mechanisms have been described in a variety of organisms. For instance, the PhoP/PhoQ regulon in Salmonella has been demonstrated to regulate transcriptional activation of surface and secretory proteins, enzymes that modify lipopolysaccharide, lipid and protein constituents of the outer membrane and proteases that likely degrade certain AMPs.
Examples of antimicrobial peptides
| Cationic peptides enriched for specific amino acids | |
|---|---|
| Glycine-containing peptides | Hymenoptaecin from honeybees |
| Glycine- and proline-containing peptides | Coleoptericin from beetles Holotricin from beetles |
| Histidine-containing peptides | Histatins from humans and some higher primates |
| Proline-containing peptides | Abaecin from honeybees |
| Proline- and arginine-containing peptides | Apidaecins from honeybees Bactenicins from cattle Drosocin from Drosophila PR-39 from pigs |
| Proline- and phenylalanine-containing peptides | Prophenin from pigs |
| Tryptophan-containing peptides | Indolicidin from cattle |
| Linear cationic α-helical peptides | |
|---|---|
| Andropin from insects Bombinin from amphibians Buforin II from amphibians CAP18 from rabbits Cepropins from insects Cecropin P1 from the pig intestinal parasitic nematode, Ascaris suum Ceratotoxin from insects Dermaseptin from amphibians LL-37 from human Magainin from amphibians Melittin from insects Pleurocidin from Pseudopleuronectes americanus |
| Anionic and cationic peptides that contain cysteine and form disulfide bonds | |
|---|---|
| 1 Disulfide bond | Brevinins |
| 2 Disulfide bonds | Protegrins from pigs |
| 3 Disulfide bonds | α-Defensins from human, rabbits and rats β-Defensins from humans, cattle, mice, rats, pigs, goats and poultry θ-Defensin from the rhesus monkey Insect defensins (Defensin-A from Aedes aegypti) |
| 4 Disulfide bonds | Antifungal defensins from plants Drosomycin from Drosophila |
| Anionic peptides | Dermcidin from human skin Maximin H5 from amphibian skin |
| Anionic and cationic peptide fragments derived from precursor proteins | Antimicrobial domains from bovine α-lactalbumin, human hemoglobin, lysozyme, and ovalbumin Aromatic dipeptides from dipteran larvae Casocidin I from human casein Enkelytin from proenkaphalin A Lactoferricin from lactoferrin |
Adapted from K.A. Brogden, Nat. Rev. Microbiol. 3, 238-250 (2005)
IMPORTANT FAMILIES OF AMPs
Bombinins
Bombinins constitute a family of AMPs produced in fire-bellied toads (Bombina species) active against Gram-negative and Gram-positive bacteria and fungi. Bombinins, bombinin-like peptides (BLPs), and Bombinin H molecules are found in the species Bombina bombina, Bombina variegata, and Bombina orientalis, whereas the homologous maximins and maximin H peptides are derived from the giant fire-bellied toad Bombina maxima. Bombinin H peptides contain either 17 or 20 amino acid residues and are more hydrophobic than bombinins, some of them contain D-alloisoleucine at position 2. They exhibit lower antibacterial activity than bombinins but, in contrast to them, they possess haemolytic activity.
Cathelicidins
Members of this family are amphipathic, cationic peptides with a broad-spectrum antimicrobial activity. Cathelicidins typically act by disrupting the integrity of bacterial membranes. They are characterized by an evolutionary conserved N-terminal cathelin- like domain of approximately 99-114 amino acid residues linked to a C-terminal antimicrobial domain of 12-100 residues that can be released upon proteolytic processing. Members of this family include linear peptides amongst them a number of proline-rich AMPs that show different types of proline repeat motifs (Bac5, Bac7, PR-39, prophenins) and the tryptophan-rich indolicidin characterized by three regularly spaced proline residues. The protegrins (PG-1 to PG-5) contain two disulfide bridges and an amidated C-terminus. Cathelicidins have been found in every mammalian species examined. In human, LL-37 (Product 4042456) is the only member of the cathelicidin family. The peptide consists of 37 amino acids and contains two leucine residues at the N-terminus. It is proteolytically cleaved from the 18 kDa precursor protein human cathelicidin antimicrobial protein CAP-18. LL-37 is primarily produced by phagocytic leucocytes and epithelial cells, and is involved in various processes such as direct killing of microorganisms, binding and neutralizing LPS, chemotaxis and chemokine induction, regulation of inflammatory responses, and wound healing. Its production is influenced by several factors such as microbial products, host cytokines, vitamin D3, and availability of oxygen. LL-37 orthologues in mouse and rat are CRAMP (mouse) (Product 4056438) and CRAMP (rat), respectively.
Cecropins
Cecropins were first isolated from the giant silk moth Hyalophora cecropia. They can form amphipathic, α-helical structures and are structurally related to other cecropins as bactericidin, lepidopteran, and sarcotoxin. Cecropin P1 (Product 4039862), found in pig intestine, also belongs to this family. Most cecropins show broad-spectrum antibacterial activity. Cecropin A (Product 4030488) and B (Product 4030477) have also been demonstrated to possess tumoricidal activity against mammalian leukemia, lymphoma, and carcinoma cell lines.
Ceratotoxins
This family consists of cationic α-helical amphipathic peptides expressed in the female reproductive accessory glands of the Mediterranean fruit fly Ceratitis capitata. The production of the peptides is enhanced by mating. Ceratotoxin A and ceratotoxin B are 29 amino acid peptides differing in two amino acids. Ceratotoxin C and D consist of 32 and 36 amino acids, respectively. The peptides of this family are active against Gram-negative as well as Grampositive bacteria and are supposed to act via the Barrel-Stave model. Ceratotoxin A has been shown to be mainly antibacterial for Gram-negative organisms.
Defensins
Defensins are small cysteine-rich cationic peptides containing three or four disulfide bridges. They have been isolated from molluscs, acari, arachnids, insects, mammals, and plants. They are further divided into families on the basis of the spatial distribution of their cysteine residues. Three families, the α-, β- and θ-defensins, can be distinguished in mammals. α- and β-defensins are characterized by antiparallel β-sheet structures stabilized by three disulfide bonds. The θ-defensins are found in rhesus monkey and some other non-human primates but not in human, chimpanzee and gorilla. They consist of two nine amino acid peptides derived from different precursor proteins joined by head-to-tail cyclization. Invertebrate and plant defensins contain three or four disulfide bridges, respectively. Insect and mammalian defensins are mainly active against bacteria while most plant defensins possess antifungal activity.
Dermaseptins
The peptides of the dermaseptin family are closely related and consist of 28-34 amino acids. They were originally isolated from skin extracts of the South American arboreal frog Phyllomedusa sauvagei and contain a conserved tryptophan residue at position 3. Dermaseptins exhibit broad-spectrum antimicrobial activity against Gram-positive and Gram-negative bacteria.
Histatins
Histatins are histidine-rich and mostly cationic peptides found in the saliva of humans and some higher primates. They are active against a broad-spectrum of bacteria and fungi. The antifungal activity of the human salivary peptide histatin-5 has been extensively studied and is supposed to be due to inhibition of mitochondrial respiration and the formation of reactive oxygen species. Histatin-5 has also been shown to inhibit both host-derived and bacterial proteolytc enzymes involved in peridontal diseases. Histatin-8, a peptide from human parotid secretion, has been shown to inhibit hemagglutination activity of Porphyromonas gingivalis 381, a Gram-negative bacterium involved in certain forms of periodontal disease. The peptide may function as a binding domain for the hemagglutinins of Porphyromonas gingivalis during agglutination.
Magainins
Magainins constitute a family of linear amphipathic cationic AMPs discovered in the skin of Xenopus laevis. The two closely related members of this family, magainin I (Product 4012844) and magainin II (Product 4013706) differ merely in two positions and are 23 amino acids in length. Magainins exhibit broad-spectrum antimicrobial activity against Gram-negative and Gram-positive bacteria, fungi and protozoa and are also cytotoxic for many murine and human cancer cell lines.
Conclusions
The structures of AMPs represent a unique source for the targeted exploration of new applications in the therapy of microbial and viral infection, cancer, and sepsis. Modern synthetic methods will allow the relatively cheap and accurate production of lead compounds and peptide candidates. The achievements in peptide library generation, analytical methods as mass spectrometry, and screening and formulation technologies may contribute to solve intrinsic problems associated with the use of AMPs as therapeutic agents such as susceptibility to proteases and host toxicity. Bachem has considerable expertise and long-standing experience in peptide synthesis. With our capacity to upscale the production of simple and modified peptides, we are the partner of choice for the pharmaceutical industries.
Download the Antimicrobial peptide white paper
Subscribe to our newsletter
"*" indicates required fields
References
E. Martin et al.
Defensins and other endogenous peptide antibiotics of vertebrates.
J. Leukoc. Biol. 58, 128-136 (1995)
D. Andreu and L. Rivas
Animal antimicrobial peptides: an overview.
Biopolymers 47, 415-433 (1998)
R.E. Hancock and D.S. Chapple
Peptide antibiotics.
Antimicrob. Agents Chemother. 43, 1317-1323 (1999)
M. Zasloff
Antimicrobial peptides of multicellular organisms.
Nature 415, 389-395 (2002)
D.A. Devine
Antimicrobial peptides in defence of the oral and respiratory tracts.
Mol. Immunol. 40, 431-443 (2003)
A.E. Shinnar et al.
Cathelicidin family of antimicrobial peptides: proteolytic processing and protease resistance.
Bioorg. Chem. 31, 425-436 (2003)
M.R. Yeaman and N.Y. Yount
Mechanisms of antimicrobial peptide action and resistance.
Pharmacol. Rev. 55, 27-55 (2003)
T. Jin et al.
Staphylococcus aureus resists human defensins by production of staphylokinase, a novel bacterial evasion mechanism.
J. Immunol. 172, 1169-1176 (2004)
M.E. Selsted
Theta-defensins: cyclic antimicrobial peptides produced by binary ligation of truncated alpha-defensins.
Curr. Protein Pept. Sci. 5, 365-371 (2004)
M. Zanetti
Cathelicidins, multifunctional peptides of the innate immunity.
J. Leukoc. Biol. 75, 39-48 (2004)
K.A. Brogden
Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria?
Nat. Rev. Microbiol. 3, 238-250 (2005)
M. Zanetti
The role of cathelicidins in the innate host defenses of mammals.
Curr. Issues Mol. Biol. 7, 179-196 (2005)
H. Jenssen et al.
Peptide antimicrobial agents.
Clin. Microbiol. Rev. 19, 491-511 (2006)
V. Nizet
Antimicrobial peptide resistance mechanisms of human bacterial pathogens.
Curr. Issues Mol. Biol. 8, 11-26 (2006)
M. Golec
Cathelicidin LL-37: LPS-neutralizing, pleiotropic peptide.
Ann. Agric. Environ. Med. 14, 1-4 (2007)
D.W. Hoskin and A. Ramamoorthy
Studies on anticancer activities of antimicrobial peptides.
Biochim. Biophys. Acta 1778, 357-375 (2008)
J. Schauber and R.L. Gallo
Antimicrobial peptides and the skin immune defense system.
J. Allergy Clin. Immunol. 122, 261-266 (2008)
H. Suttmann et al.
Antimicrobial peptides of the cecropin-family show potent anti-tumor activity against bladder cancer cells.
BMC Urol. 8, 5 (2008)
K. Yamasaki and R.L. Gallo
Antimicrobial peptides in human skin disease.
Eur. J. Dermatol. 18, 11-21 (2008)
G. Diamond et al.
The roles of antimicrobial peptides in innate host defense.
Curr. Pharm. Des. 15, 2377-2392 (2009)
Y. Lai and R.L. Gallo
AMPed up immunity: how antimicrobial peptides have multiple roles in immune defense.
Trends Immunol. 30, 131-141 (2009)
A. Nijnik and R.E. Hancock
The roles of cathelicidin LL-37 in immune defences and novel clinical applications.
Curr. Opin. Hematol. 16, 41-47 (2009)
M. Otto
Bacterial sensing of antimicrobial peptides.
Contrib. Microbiol. 16, 136-149 (2009)
R. Palffy et al.
On the physiology and pathophysiology of antimicrobial peptides.
Mol. Med. 15, 51-59 (2009)
M. Simmaco et al.
Bombinins, antimicrobial peptides from Bombina species.
Biochim. Biophys. Acta 1788, 1551-1555 (2009)
A. Zairi et al.
Dermaseptins and magainins: antimicrobial peptides from frogs’ skin-new sources for a promising spermicides microbicides-a mini review.
J. Biomed. Biotechnol. 2009, 452567 (2009)
P. Mendez-Samperio
The human cathelicidin hCAP18/LL-37: a multifunctional peptide involved in mycobacterial infections.
Peptides 31, 1791-1798 (2010)
J. Wiesner and A. Vilcinskas
Antimicrobial peptides: the ancient arm of the human immune system.
Virulence 1, 440-464 (2010)
C.D. Fjell et al.
Designing antimicrobial peptides: form follows function.
Nat. Rev. Drug Discov. 11, 37-51 (2012)
K. Parn et al.
The antimicrobial and antiviral applications of cell-penetrating peptides.
Methods Mol. Biol. 1324, 223-245 (2015)
