MEET US AT CPHI JAPAN

Bachem Japan KK is participating in the CPhI Japan 2019! More than 20’000 pharma professionals from more than 65 countries are expected in Japan, the third largest pharmaceutical industrial nation worldwide, to attend this exciting pharmaceutical event.
The 2019 edition takes place on March 18 – 20, 2019 in the Big Sight Exhibition Center in Tokyo, together with the co-located ICSE (contract manufacturing and services), P-MEC (machinery, technology & equipment), BioPh (biopharmaceutical developments), InnoPack (packaging & drug delivery systems), FDF (finished dosage formulation) and, new for 2019, NEX (natural extracts) and Pharma IT & Digital Expo shows.
We kindly invite you to drop by our Booth #S29. We will be delighted to discuss with you how Bachem can meet your API custom manufacturing needs. With our capacity to produce generic peptide API in quantities of hundreds of kilograms and small molecules in tens of tons per year and our record of over 80 DMF filings in the pipeline, we will certainly be able to support the success of your projects from initiation, through all clinical development phases to commercial supply. Please contact us to schedule a meeting in advance.
We look forward to meeting you at CPhI Japan 2019!
PEPTIDES AND OSTEOPOROSIS

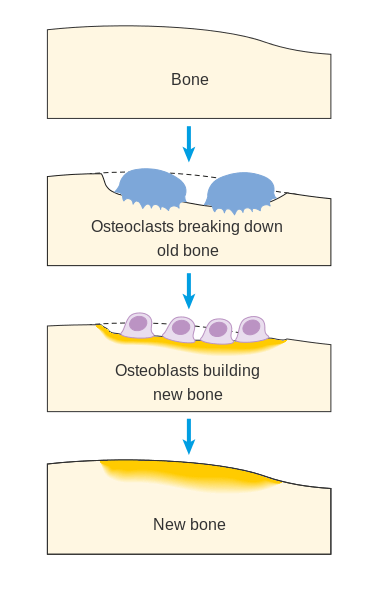
Over 200 million people worldwide suffer from osteoporosis, the disease in which bone density and quality of bone are reduced causing bones to become more porous and fragile. Globally, approximately one in three women and one in five men are at risk for having an osteoporotic fracture. During normal bone maintenance, bones undergo a process called remodeling in which bone cells dissolve, known as resorption, and new bone cells deposit osteoid, known as formation. Remodeling (Figure 1) involves cells that resorb bone called osteoclasts and cells that form bone called osteoblasts. In people with osteoporosis, the amount of bone loss is greater than the growth of new bone causing bones to weaken and become susceptible to fracture (1). Until recently, this negative balance in bone formation and resorption has been treated with anti-resorptive drugs that inhibit osteoclast formation and/or activity. Over the past couple of decades new treatment approaches with anabolic, or bone building agents, have emerged offering new promise for people with osteoporosis (2). Peptides involved in bone processes are playing important roles as treatment options and as potential diagnostic indicators of osteoporosis.
Figure 1 Bone Remodeling
(Cancer Research UK / Wikimedia Commons)
PTH Peptides
Parathyroid hormone (PTH) and parathyroid hormone-related peptide (PTHrP) have been at the center of research and development efforts towards finding anabolic treatments for osteoporosis.
PTH is an 84 amino acid peptide that is secreted by the parathyroid glands in response to lowered calcium. It acts to increase renal tubular reabsorption of calcium and stimulates renal calcitriol (1,25-dihydroxyvitamin D3) production to indirectly increase intestinal calcium absorption and regulate bone remodeling. The ligand for PTH is the PTH-1 receptor, a G protein-coupled receptor that is expressed in kidney and bone. PTH acts to stimulate bone formation by increasing the number and action of osteoblasts and decreasing osteoblast cell death. PTH also promotes the building of bone by regulating skeletal growth factors such as IGF-1 and growth factor antagonists such as sclerostin (3).
PTHrP is expressed in normal tissues throughout development. PTHrP, like PTH, binds to the PTH-1 receptor. PTHrP is involved in the regulation of cellular growth, differentiation, development and cell death. It is also involved in epithelial calcium transport in a variety of tissues. In studies, PTHrP was shown to be required for normal bone development (3).
While PTH (1-34) and PTH (1-84) stimulate bone formation via osteoblasts, both peptides also stimulate osteoclast-dependent bone breakdown to a smaller degree. Ideally, a treatment for osteoporosis would stimulate bone formation without stimulating any resorption (3).
Calcitonin
Calcitonin (salmon) is a peptide composed of 32 amino acids that is naturally secreted by cells in the thyroid gland. Calcitonin binds to the calcitonin receptor that is found in osteoclasts and acts to enhance the production of vitamin D producing enzymes. Accordingly, calcitonin results in increased calcium retention and bone density (4).
Osteocalcin
During bone remodeling, osteoblasts secrete osteocalcin, a bone formation biomarker, during the bone formation phase. Osteocalcin has a strong affinity for hydroxyapatite, the main inorganic component of the bone matrix. In osteoporosis, there is decreased hydroxyapatite crystal formation, which results in increased serum levels of osteocalcin. Serum osteocalcin levels may be useful for the diagnosis of osteoporosis and osteopenia, a condition of low bone density that can be a precursor to osteoporosis (5).
Osteoporosis Treatment
Osteoporotic fracture can lead to pain, impaired mobility and poor quality of life but fortunately, osteoporosis is treatable and patients can avoid fracture through medical treatment combined with lifestyle changes. As first-line treatment, patients are often treated with anti-resorptive drugs that preserve bone called bisphosphonates, the most common of which is alendronate. Bisphosphonates inhibit the recruitment of osteoclasts and work by inhibiting the osteoclast-stimulating activity of osteoblasts (2). In some countries, anabolic therapies are also available for the treatment of osteoporosis including PTH and PTHrP analog drugs.
Calcitonin (Fortical®, Miacalcin®, Calcimar®) is an anti-resorptive drug that is commercially available for the treatment of osteoporosis. Studies have shown that calcitonin decreases the occurrence of vertebral fractures but there is no evidence that calcitonin decreases the risk of non-vertebral fractures. Due to the availability of more effective drugs, calcitonin is not a first-line treatment for osteoporosis (6).
Eli Lilly and Company’s Forteo® (PTH (1-34), teriparatide) was the first drug approved by the U.S. Food and Drug Administration (FDA) for treating osteoporosis. Forteo works mainly by increasing bone formation and the drug is commercially available in many countries across the world. Forteo consists of the first thirty-four amino acids of human PTH. In clinical trials, PTH (1-34) demonstrated a 65% risk reduction in vertebral fractures and a 53% risk reduction in non-vertebral fractures compared to placebo in postmenopausal women with osteoporosis. Due to concerns that prolonged PTH (1-34) treatment could cause osteosarcomas, PTH (1-34) has a maximum treatment duration of 24 months. In addition, studies have shown that after the first year of treatment, bone turnover markers begin to plateau and then decline (3).
In Europe, Preotact® (PTH (1-84)) was approved for the treatment of osteoporosis but the marketing authorization holder withdrew it later. In clinical trials, PTH (1-84) showed a 58% reduction in new or worsened vertebral fractures in postmenopausal women with osteoporosis. In 2015, the FDA approved Natpara® (PTH 1-84) to control hypocalcemia in patients with hypoparathyroidism and in 2017, the European Medicines Agency granted marketing authorization to Natpar® (PTH 1-84) for the treatment of patients with chronic hypoparathyroidism (4).
In April 2017, the FDA approved a new anabolic agent, Tymlos® (abaloparatide). Radius Health’s Tymlos is indicated for the treatment of postmenopausal women with osteoporosis at risk for fracture. Tymlos is a novel PTHrP (1-34) analog that regulates cell proliferation and differentiation in developing tissues without stimulating bone resorption and hypercalcemia to the same level as PTH (4). In clinical studies, Tymlos has been shown to decrease incidence of new vertebral and non-vertebral fractures, to increase bone mineral density (BMD) and increase a marker of bone formation. In the ACTIVE trial (Abaloparatide Comparator Trial In Vertebral Endpoints), abaloparatide achieved an 86% reduction in risk of new vertebral fractures and a 43% decrease in risk of non-vertebral fractures in postmenopausal women with osteoporosis as compared to the placebo. The FDA approval of Tymlos was based on 18 months of results from the ACTIVE clinical trial and Tymlos use is limited to 18 months (4).
In the research and development pipeline, other peptides are under investigation as potential anabolic treatments for osteoporosis such as PTHrP (1-36) and a PTHrP (1-34) analog (3). PTHrP binds to the PTH-1 receptor and activates signal transduction equally to PTH. In a clinical trial of daily synthetic human PTHrP (1-36), markers of bone formation were increased while bone resorption markers were unchanged suggesting that PTHrP (1-36) might have a “pure” anabolic effect on bone (3).
Outlook
Peptides such as PTH and PTHrP analogs are likely to continue to play an important role in osteoporosis management. While bisphosphonates have been the first-line treatment option, there is concern about their long-term safety. New treatments with novel mechanisms to treat osteoporosis are expected in the future. To support advancements in the field of osteoporosis research, Bachem offers a wide selection of peptides.
References
(1) What is osteoporosis? International Osteoporosis Foundation (2017)
(2) T.K.Martin, Bone biology and anabolic therapies for bone: Current status and future prospects. J. Bone Metab. 21, 8-20 (2014)
(3) M.Augustine, M. Horowitz, Parathyroid hormone and parathyroid hormone-related protein analogs as therapies for osteoporosis. Curr. Osteoporos. Rep. 11, 400-406 (2013)
(4) GlobalData (2018)
(5) S.Singh et al., Serum osteocalcin as a diagnostic biomarker for primary osteoporosis in women. J. Clin. Diagn. Res. 9, RC04-RC07 (2015)
(6) GM.Jeremiah et al., Diagnosis and management of osteoporosis. Am. Fam. Physician. 92, 261-268 (2015)
PEPTIDES IN CLINICAL DEVELOPMENT FOR OSTEOPOROSIS
Osteoporosis is a common metabolic bone disease that is estimated to cause more than 8.9 million fractures worldwide annually (1). The main goal of osteoporosis treatment is to prevent fragility fractures that are most frequent in the hip, spine and wrist. Current treatment of osteoporosis is focused on anti-resorptive agents. In the US, there are two FDA-approved anabolic therapies for osteoporosis: abaloparatide (Tymlos®, Radius Health) and teriparatide (Forteo®, Eli Lilly). As shown in Table 1, there are several peptide therapeutics in clinical development for the treatment of osteoporosis. Some product candidates in the development pipeline are biosimilars and others are using new delivery technologies in order to improve anabolic therapy and increase patient compliance.
| Product Name | Active Ingredient | Condition Treated | Highest Phase | Company |
|---|---|---|---|---|
| TBRIA | Calcitonin (salmon) | Postmenopausal Osteoporosis | Pending Approval | Enteris BioPharma Inc |
| Uni-PTH | Teriparatide | Postmenopausal Osteoporosis | Pending Approval | Uni-Bio Science Group Ltd |
| PF-708 | Teriparatide | Osteoporosis | III | Pfenex Inc |
| ENZ101 | Teriparatide | Postmenopausal Osteoporosis | III | Enzene Biosciences Ltd |
| MicroCor hPTH (1-34) | Teriparatide | Osteoporosis | II | Corium International |
| EB-613 | Teriparatide | Osteoporosis | I | Entera Bio Ltd |
| PTH (1-84) | --- | Postmenopausal Osteoporosis | I | Shandong Danhong Pharmaceutical Co Ltd |
| SBL-001 | Teriparatide | Osteoporosis | I | Stelis Biopharma Pvt Ltd |
Pending Approval
Enteris BioPharma is developing TBRIATM as an oral form of calcitonin for the treatment of postmenopausal osteoporosis. Calcitonin inhibits bone resorption and promotes bone formation by osteoblasts. The product uses the company’s Peptelligence® oral delivery technology. In 2015, an NDA for TBRIA was submitted to the U.S. Food and Drug Administration (FDA) (2). In 2018, R-Pharm JSC acquired the development rights to TBRIA from Tarsa Therapeutics, the original development partner for TBRIA. R-Pharm will be responsible for any further development and subsequent regulatory submissions (3).
Uni-Bio Science Group is developing teriparatide for the treatment of osteoporosis in China. In 2015, the company filed a New Drug Applications (NDA) with the China Food and Drug Administration (CFDA) for Uni-PTH in a powder formulation. The company is also developing a liquid formulation of teriparatide (2).
Phase III
PF-708 (teriparatide) is under development at Pfenex for the treatment of osteoporosis. In 2018, Pfenex completed a Phase III study that compared the effects of PF708 and Forteo in patients with osteoporosis. The company anticipates a potential commercial launch of the product in the US by the fourth quarter of 2019 (2).
Enzene Biosciences is developing ENZ101, a teriparatide biosimilar. The product is in Phase III development for the treatment of postmenopausal osteoporosis. In 2018, the company registered a Phase III study with the Clinical Trials Registry of India. The study will compare the safety and efficacy of teriparatide versus Forteo in the treatment of postmenopausal osteoporosis (2).
Phase II
Corium International is developing MicroCor hPTH (1-34), a teriparatide product candidate that is administered via transdermal delivery. This teriparatide product candidate employs the company’s MicroCor technology that is based on microneedles, or dissolving microstructures, for the delivery of molecules such as peptides (4). In 2017, Corium International announced that the company had completed a Phase IIa study of MicroCor hPTH (1-34) for the treatment of osteoporosis (2).
Phase I
Entera Bio is developing EB-613 (teriparatide), an oral form of teriparatide. The drug candidate is being developed based on Entera’s platform technology that increases drug half-life and bioavailability (2). The technology utilizes a combination of a synthetic absorption enhancer to facilitate enhanced absorption and protease inhibitors to prevent degradation (5). In 2019, the company is planning to start a Phase IIa dose-ranging study of EB-613 in patients with osteoporosis (2).
PTH (1-84) recombinant peptide is under development at Shandong Danhong Pharmaceutical for the treatment of postmenopausal osteoporosis. The peptide acts as a parathyroid receptor agonist. In 2018, the company registered a Phase I trial of PTH (1-84) with the Chinese Clinical Trial Registry (2).
Stelis Biopharma is developing SBL-001, a teraparatide biosimilar, for the treatment of osteoporosis. In 2018, the company completed a comparative study that evaluated the pharmacokinetics, safety and tolerability of SBL-001 with Forsteo® (teriparatide, Eli Lilly). The company plans to launch the product in 2019 (2).
Conclusion
New treatment options for osteoporosis have emerged in recent years and more can be expected in the future. To support researchers and organizations involved in the field of osteoporosis research and drug development, Bachem offers custom peptide synthesis servic, the production of New Chemical Entities and Generic Active Pharmaceutical Ingredients (API). Bachem also offers a wide selection of peptides for osteoporosis research.
References
(1) Facts and Statistics. International Osteoporosis Foundation (2018)
(2) GlobalData (2018)
(3) R-Pharm JSC acquires rights to Tarsa’s TBRIA™, novel oral treatment for post-menopausal osteoporosis. Tarsa Therapeutics (2018)
(4) MicroCor. Corium (2018)
(5) Oral Drug Delivery. EnteraBio (2018)
MEET BACHEM: NINA ULLRICH
What is your official job title at Bachem?
Sales Representative Inside Sales
How long have you been with Bachem? Where did you work before Bachem?
I started working at Bachem in June 2018. Before joining Bachem I worked in the Marketing & Sales department of a Swiss food company.
What is your academic background?
I studied Ecotrophology at Justus-Liebig University in Giessen, Germany. Afterwards I gained my M.Sc. in Nutritional Sciences at ETH Zürich.
What do you like to do outside of work?
I like playing tennis in summer and in the winter season I go jogging. I also enjoy cooking and spending time with my friends and family.
What makes a perfect day for you?
Waking up on a sunny day by the smell of freshly squeezed orange juice and croissants. During the day there are a lot of possibilities how to make the day perfect: hiking, listening to the sea, staying outside and enjoying the nature. The perfect day ends with delicious food and some wine.
Thank you very much Nina.

Peptide highlights
Interesting news about peptides in basic research and pharmaceutical development:
Receptor structure could be key to developing new osteoporosis drugs-News-Medical.Net
Cancer vaccines: using neo-antigen peptide array screening-GEN
Scorpion venom to shuttle drugs into the brain-Science Daily
Targeting MC1R in metastatic melanoma-University of Colorado Cancer Center
LITERATURE CITATIONS
Bachem peptides and biochemicals are widely cited in research publications. Congratulations to all our customers with recent publications!
J.Delgado-Calle et al.
MMP14 is a novel target of PTH signaling in osteocytes that controls resorption by regulating soluble RANKL production.
The FASEB Journal 32, 2878-2890 (2018)
A.Makino et al.
Abaloparatide exerts bone anabolic effects with less stimulation of bone resorption-related factors: A comparison with teriparatide.
Calcified Tissue International 103, 289-297 (2018)
Y.Wu et al.
Mettl3-mediated m6A RNA methylation regulates the fate of bone marrow mesenchymal stem cells and osteoporosis.
Nature Communications 9, 4772 (2018)
L.Xiao et al.
FGF2 crosstalk with Wnt signaling in mediating the anabolic action of PTH on bone formation.
