NESFATIN
Nesfatin Peptides and Precursor Protein
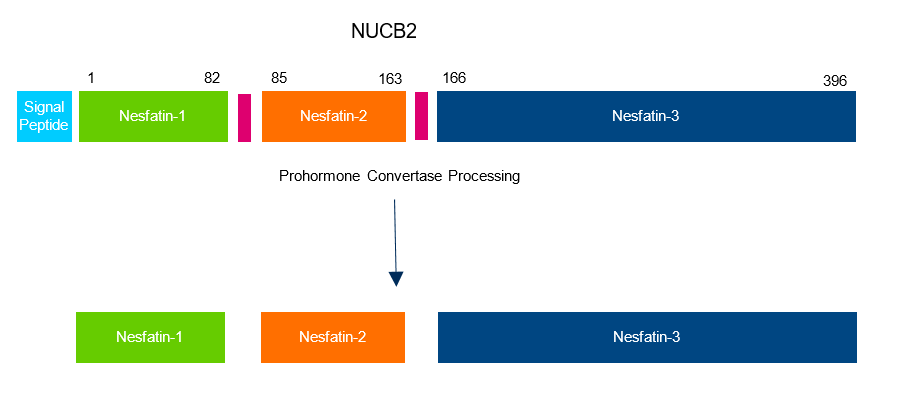
Nesfatin-1 is an 82 amino acid peptide that is derived from the 396 amino acid precursor protein nucleobindin 2 (NUCB2). NUCB2 is expressed in the central nervous system (CNS) as well as in peripheral tissues. The protein is processed by pro-hormone convertase-1/3 and the resulting peptides are nesfatin-1 (amino acids 1-82), nesfatin-2 (amino acids 85-163) and nesfatin-3 (amino acids 166-396) as shown in Figure 1. Nesfatin-1 is the active sequence of nesfatin and exerts an anorexigenic effect (3). The 29 amino acid fragment of nesfatin-1, nesfatin-1 (30-59), known as the active core sequence of nesfatin-1, has also demonstrated biological activity.
Figure 1. Three major peptide products are derived from proteolytic processing of NUCB2 including the active peptide nesfatin-1 and the two derivatives, nesfatin-2 and -3 with unknown functions.
Feeding
Studies in rodents and goldfish have demonstrated that nesfatin-1 injected into the brain elicits an anorexigenic response. Centrally injected nesfatin-1 reduces the nocturnal food intake in mice by reducing meal size and increasing intervals between meals (1). The application of nesfatin-2 or -3 does not produce this same effect on feeding. The action of nesfatin-1 was found to occur through a leptin-independent mechanism since nesfatin-1 had an anorexigenic action in Zucker rats (4), which do not have functional leptin (4). Central nesfatin-1 is thought to mediate its anorexigenic effect via activation of melanocortin 3/4 and corticotropin releasing factor (CRF)-2 signaling and also by hyperpolarizing neurons containing neuropeptide Y (5). In addition, intracerebroventricular (icv) injection of nesfatin-1 in mice and rats inhibits gastric emptying and reduces gastroduodenal motility in mice. Nesfatin-1 also inhibits gastric acid secretion via a central vagal mechanism in rats. Also, there is evidence for a role of nesfatin-1 in the control of body fluid regulation (6).
Blood Glucose Homeostasis
In rodents, dogs, pigs and humans, nesfatin-1 has been found in the pancreatic islets co-localized with insulin. In a study to characterize the effects of nesfatin-1 on insulin secretion, nesfatin-1 stimulated glucose-induced insulin release from cultured MIN6 cells. In contrast, no significant difference in insulin release was found in the presence of low glucose. In addition, nesfatin-1 stimulated glucose-induced insulin release from mouse islets (7).
Depression and Anxiety
Nesfatin-1 may be involved in depression and mediating anxiety and fear related responses. It has been demonstrated that nesfatin-1 and two energy regulatory hormones, acylated and des-acylated ghrelin, are significantly higher in patients with moderate and severe major depression disorders and are associated with the severity of the depression (8). In rats, nesfatin-1 was shown to increase anxiety and fear-related behaviors such as a decreased percentage of time spent on open arms in an elevated maze and increased fear-potentiated startle response following icv injection of nesfatin-1 (9).
Sleep
Sleep disorders affect millions of people and are often a main symptom of depression. Patients with major depressive disorder possess high plasma levels of nesfatin-1. Investigators have examined the potential connection between nesfatin (mRNA and protein) expression and sleep. In rat studies, REM sleep deprivation resulted in reduced expression of nesfatin mRNA and protein expression while REM sleep rebound reversed this. In addition, central administration of nesfatin-1 decreased the time spent in REM sleep and intermediate stage of sleep (IS) (10).
Cardiovascular
Several of the peptides that influence feeding such as orexin, adrenomedullin and α-melanocyte-stimulating hormone (α-MSH), also have an impact on cardiovascular function. Researchers have been interested in exploring the potential influence of nesfatin-1 on cardiovascular actions. In rat studies, researchers tested the ability of nesfatin-1 to affect mean arterial pressure (MAP) and found that icv administration of nesfatin-1, in doses that inhibit feeding, resulted in significant increases in MAP in conscious freely moving rats (11).
Reproduction
Research in female rats has demonstrated that nesfatin-1 may play a key role in the control of female puberty. During female puberty transition, Nucb2 mRNA and protein expression were significantly increased in the hypothalamus. Conditions of negative energy balance such as fasting or subnutrition, decreased Nucb2 mRNA and protein expression in the hypothalamus. In addition, central nervous system administration of nesfatin-1 induced luteinizing hormone secretion in freely feeding and short-term fasted pubertal female rats. Conversely, administration of nesfatin-1 did not cause any detectable changes in the circulating levels of gonadotropins in adult female rats (12).
Conclusion
Nesfatin-1 plays roles in several areas such as feeding, depression and anxiety, cardiovascular system, sleep and reproduction. To support life scientists and researchers studying the roles of nesfatin-1 in these systems and processes, Bachem offers a selection of nesfatin peptides in addition to a comprehensive custom synthesis service.
References
1) A.Stengel and Y.Tache. Minireview: Nesfatin-1—an emerging new player in the brain-gut, endocrine, and metabolic axis. Endocrinology 152(11), 4033-4038 (2011)
2) R.Dore et al. Nesfatin-1: functions and physiology of a novel regulatory peptide. Journal of Endocrinology 32(1), R45-R65 (2017)
3) M.A.Schalla and A.Stengel. Current understanding of the role of nesfatin-1. Journal of the Endocrine Society 2, 1188-1206 (2018)
4) S.Oh-I et al. Identification of nesfatin-1 as a satiety molecule in the hypothalamus. Nature 443, 709-712 (2006)
5) A.Stengel, M.Goebel and Y.Taché. Nesfatin-1: a novel inhibitory regulator of food intake and body weight. Obesity Reviews 12(4), 261-271 (2011)
6) A.Stengel, M.Mori and Y.Taché. The role of nesfatin-1 in the regulation of food intake and body weight – recent developments and future endeavors. Obesity Reviews 14(11), 859-870 (2013)
7) R.Gonzalez et al. Nesfatin-1 exerts a direct, glucose-dependent insulinotropic action on mouse islet β- and MIN6 cells. Journal of Endocrinology 208(3), R9-R16 (2011)
8) S.Algul and O.Ozcelik. Major depressive disorders. Psychiatry Investigation 15(2), 214-218 (2018)
9) Z.Merali et al. Nesfatin-1 increases anxiety- and fear-related behaviors in the rat. Psychopharmacology (Berl). 201(1), 115-123 (2008)
10) S.Vas. Nesfatin-1/NUCB2 as a potential new element of sleep regulation in rats. PLoS One 8(4), p. e59809 (2013)
11) G.L.C.Yosten and W.K.Samson. Nesfatin-1 exerts cardiovascular actions in brain: possible interaction with the central melanocortin system. American Journal of Physiology 297(2), R330-R336 (2009)
12) D.Garcia-Galiano et al. The anorexigenic neuropeptide, nesfatin-1, is indispensable for normal puberty onset in the female rat. Journal of Neuroscience 30(23), 7783-7792 (2010)
ANTI-OBESITY PEPTIDES IN CLINICAL DEVELOPMENT
Obesity is a serious issue with the worldwide prevalence of obesity almost tripling between 1975 and 2016 according to the World Health Organization (1). In addition, the increase of overweight and obese children and adolescents is of concern with the prevalence rising to just over 18% among this age group in 2016 (1). Obesity can lead to health conditions such as heart disease, stroke, type 2 diabetes and some types of cancer. Due to the limitations of lifestyle interventions and bariatric surgery for obesity, there is an urgent need for drug treatments. The peptide hormones involved in regulating food intake, energy expenditure and glucose homeostasis, along with their agonists and antagonists, are becoming important tools in the search for anti-obesity agents. Several peptide drug candidates are currently undergoing development for the treatment of obesity as shown in Table 1.
| Product Name | Company or Organization | Highest Phase | Target |
|---|---|---|---|
| Setmelanotide | Rhythm Pharmaceuticals | Pre-registration | Melanocortin Receptor 4 (MC4R) |
| Efpeglenatide | Sanofi | Phase III | Glucagon Like Peptide 1 Receptor (GLP1R) |
| Semaglutide (NN-9536) | Novo Nordisk AS | Phase III | Glucagon Like Peptide 1 Receptor (GLP1R) |
| Tirzepatide | Eli Lilly | Phase III | Gastric Inhibitory Polypeptide Receptor (Glucose Dependent Insulinotropic Polypeptide Receptor or GIPR); Glucagon Like Peptide 1 Receptor (GLP1R) |
| Cotadutide | AstraZeneca Plc | Phase II | Glucagon Like Peptide 1 Receptor (GLP1R); Glucagon Receptor (GL R or GCGR) |
| NN-9838 | Novo Nordisk | Phase II | Calcitonin Receptor (CALCR); Receptor Activity Modifying Protein 1 (Calcitonin Receptor Like Receptor Activity Modifying Protein 1 or RAMP1); Receptor Activity Modifying Protein 2 (Calcitonin Receptor Like Receptor Activity Modifying Protein 2 or CRLR Activity Modifying Protein 2 or RAMP2); Receptor Activity Modifying Protein 3 (Calcitonin Receptor Like Receptor Activity Modifying Protein 3 or RAMP3) |
| PB-119 | PegBio Co Ltd | Phase II | Glucagon Like Peptide 1 Receptor (GLP1R) |
| Pegapamodutide | OPKO Health Inc | Phase II | Glucagon Like Peptide 1 Receptor (GLP1R); Glucagon Receptor (GL R or GCGR) |
| Setmelanotide ER | Rhythm Pharmaceuticals | Phase I | Melanocortin Receptor 4 (MC4R) |
| G-3215 | Imperial College London | Phase I | |
| GT-001 | Gila Therapeutics | Phase I | Neuropeptide Y Receptor Type 2 (NPY Y2 Receptor or NPY2R) |
| HM-15136 | Hanmi Pharmaceuticals | Phase I | Glucagon Receptor (GL R or GCGR) |
| HM-15211 | Hanmi Pharmaceuticals | Phase I | Gastric Inhibitory Polypeptide Receptor (Glucose Dependent Insulinotropic Polypeptide Receptor or GIPR); Glucagon Like Peptide 1 Receptor (GLP1R); Glucagon Receptor (GL R or GCGR) |
| NN-9277 | Novo Nordisk | Phase I | Glucagon Like Peptide 1 (GLP1); Glucagon Receptor (GL R or GCGR) |
| NN-9423 | Novo Nordisk | Phase I | Gastric Inhibitory Polypeptide Receptor (Glucose Dependent Insulinotropic Polypeptide Receptor or GIPR); Glucagon Like Peptide 1 Receptor (GLP1R); Glucagon Receptor (GL R or GCGR) |
| NN-9775 | Novo Nordisk | Phase I | Peptide YY (PYY I or Peptide Tyrosine Tyrosine or PYY) |
| Y-242 | Zihipp Ltd | Phase I | |
| ZP-4982 | Zealand Pharma | Phase I | Islet Amyloid Polypeptide (Amylin or Diabetes Associated Peptide or Insulinoma Amyloid Peptide or IAPP) |
Advanced Candidates
Setmelanotide, a melanocortin 4 receptor (MC4R) agonist, is being developed by Rhythm Pharmaceuticals for the treatment of rare genetic disorders of obesity such as pro-opiomelanocortin (POMC) deficiency obesity and leptin receptor (LEPR) deficiency obesity. The peptide is designed to restore the function of an impaired MC4R pathway, a pathway that regulates energy expenditure and appetite (2). In May 2020, Rhythm completed its rolling submission of a New Drug Application to the U.S. Food and Drug Administration (FDA) for setmelanotide (3).
Sanofi is developing efpeglenatide, a conjugate of CA Exendin-4 and recombinant human immunoglobulin Fc fragment (LAPS-carrier), for the treatment of obesity. In 2015, Hanmi Pharmaceuticals licensed the exclusive development and commercialization rights for efpeglenatide to Sanofi. In May 2020, Hanmi announced that Sanofi intends to return all rights for efpeglenatide to Hanmi Pharmaceuticals due a change in Sanofi’s diabetes strategy (3).
Semaglutide (NN-9536), a glucagon-like peptide 1 receptor (GLP1R) agonist, is under development by Novo Nordisk for the treatment of obesity (2). In May 2020, Novo Nordisk reported positive results from a Phase IIIa trial of semaglutide exploring sustained weight management with semaglutide vs placebo. Semaglutide 2.4 mg demonstrated superior and sustained weight loss versus placebo and a 17.4% weight loss after 68 weeks in the trial (4).
Eli Lilly also has a drug candidate, tirzepatide, in development for the treatment of obesity. Tirzepatide is a dual gastric inhibitory polypeptide (GIP) and GLP1 receptor agonist. In 2019, the company registered a Phase III trial to evaluate the efficacy and safety of tirzepatide in participants without type 2 diabetes who have obesity or are overweight (4).
Conclusion
Peptides play a promising role in the fight against obesity. To support companies and organizations working in the area of obesity, Bachem offers a wide array of obesity research peptides, custom peptide synthesis, production of new chemical entities and generic active pharmaceutical ingredients.
References
1) Obesity and overweight. World Health Organization 2020.
2) GlobalData 2020.
3) Rhythm Pharmaceuticals announces FDA acceptance of new drug application for setmelanotide for the treatment of POMC and LEPR deficiency obesities. Rhythm Pharmaceuticals 2020.
4) G.Lee, Sanofi mulling a return of efpeglenatide rights to Hanmi. BioWorld 2020.
5) Semaglutide 2.4 mg demonstrates superior and sustained weight loss versus placebo and in addition a 17.4% weight loss after 68 weeks in STEP 4 trial. GlobeNewswire 2020.
6) A study of tirzepatide (LY3298176) in participants with obesity or overweight (SURMOUNT-1). ClinicalTrials.gov 2020.
MEET BACHEM: FEDERICA DECÒ
I work as Business Development Manager for New Chemical Entities (NCE) projects.
How long have you been with Bachem? Where did you work before Bachem?
I joined Bachem in August 2019. Previously I worked in the Research Center of a big chemical company as Product Development Specialist. Afterwards I worked as Food Quality Assurance Specialist in an Italian food company.
Briefly, what do you do at Bachem?
Federica: My position at Bachem consists of building long-term business relationships with existing and new customers and establishing a good client communication. Concomitantly, I collaborate internally to ensure timely business processes.
What is your academic background/degrees or training?
I am a Food Technologist by training. I did my Bachelor in Food Science & Technology at the University of Parma in Italy and obtained my Master degree at Wageningen University (The Netherlands) specialized in Ingredients Functionality.
What do you like to do outside of work?
I enjoy spending time outside with my friends, relaxing at the park. I like doing sports in the nature such as hiking and skiing during winter. Besides that, I am learning German.
What makes a perfect day for you?
A day that begins with a smile and gets over with it!
What is your business motto?
Not be a simple seller, but a person who understands customers’ needs and expectations.
What do you like most about your job?
I like my job because I am part of a dynamic team, with a key role in building business and human relationship.
Thank you very much Federica.

Peptide highlights
Interesting news about peptides in basic research and pharmaceutical development:
Peptide drugs braced for rigors of oral route-Genetic Engineering & Biotechnology News
Snakes alive: Venom may play a role in the fight against COVID-19-Monash University
A boost for cancer immunotherapy-Science Daily
HKBU-led research team develops novel antiviral targeted drug for nasopharyngeal cancer-EurekAlert!
LITERATURE CITATIONS
Bachem peptides and biochemicals are widely cited in research publications. Congratulations to all our customers with recent publications!
W.Bahlouli et al.
Stress-induced intestinal barrier dysfunction is exacerbated during diet-induced obesity.
The Journal of Nutritional Biochemistry 81, 108382 (2020)
CA.Camacho-Ramírez et al.
The leading role of peptide tyrosine tyrosine in glycemic control after Roux-en-Y gastric bypass in rats.
Obesity Surgery 30, 697-706 (2020)
P.B.Marangon et al.
Perinatal over- and underfeeding affect hypothalamic leptin and ghrelin neuroendocrine responses in adult rats.
Physiology & Behavior 215, 112793 (2020)
Y.Maejima et al.
The hypothalamus to brainstem circuit suppresses late-onset body weight gain.
