MEET US AT THE 10TH INTERNATIONAL PEPTIDE SYMPOSIUM

Join us at the 10th International Peptide Symposium (10th IPS), in conjunction with the 55th Japanese Peptide Symposium (55th JPS), on December 3 – 7, 2018, to be held in Kyoto, Japan.
Keeping in tradition, the symposium will cover a wide spectrum of topics in peptide science, including discovery, synthesis, biological activity, relationship to physiology and pathology, and application to drugs and materials. Proceedings will include lectures from internationally distinguished speakers as well as oral and poster presentations.
Do not miss the Bachem presentation at the Luncheon Seminar (12:45 – 1:25 pm): Dr. Alexander Kleinsmann, Project Chemist at Bachem AG, will present “Highly pure Octreotide – Development and Scale-Up of a State-of-the-art SPPS Process”.
Bachem supports its customers in the pursuit of groundbreaking discoveries that further scientific advances, particularly in the field of medicine. A comprehensive catalog of biochemicals deliverable ex-stock, an exclusive custom syntheses service for research labs and a full range of services to the pharma and biotech industries complete our service portfolio.
We invite you to visit us at our Booth #11: we will be delighted to learn your needs for peptides and discuss how Bachem can assist to advance your research. Please contact us to schedule a meeting in advance.
We look forward to meeting you at the IPS and JPS in Kyoto!
HISTONE PEPTIDES
The chromosomes of eukaryotes consist of chromatin, a complex formed by DNA molecules and proteins called histones. In order to bind efficiently to the negatively charged DNA via electrostatic interactions, histones need to be charged positively. Therefore, these peptides are especially rich in the basic amino acids lysine and arginine. In chromatin, the double helix structure of DNA wraps around a core of histones, with certain areas of the histones protruding from the formed “bead”, which is called a nucleosome (see Fig. 1).
Fig. 1 X-Ray Structure of the Nucleosome Core Particle, NCP147, at 1.9 Å Resolution (PDB: 1KX5).
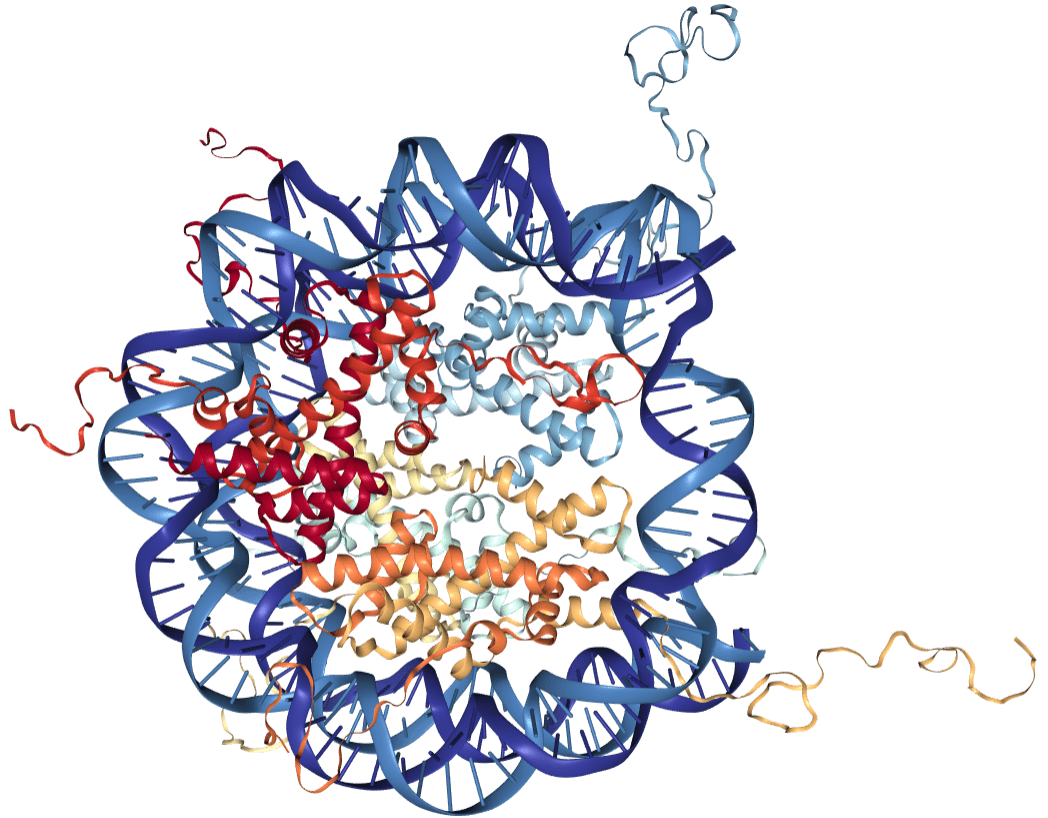
As illustrated in Fig. 1, there are varieties of different histones, which are represented by different colors within the core of the nucleosome. The various histones can be organized in five different families: H2A, H2B, H3, H4 and H1/H5, which in turn can be grouped into core histones (H2A, H2B, H3 and H4) and linker histones (H1 and H5). As the name suggests, core histones form the central part of the nucleosome, whereas linker histones connect the nucleosomes to form the so-called “Beads-on-a-String”-structure.
Post-translational modifications
Chromatin is not a rigid structure, but is rather flexible. If chromatin is in a loose conformation, the chromosomal DNA can be transcribed. Conversely, if chromatin is condensed (creating a complex called heterochromatin), it is inactive, and DNA transcription does not occur. An important factor governing the structure of chromatin is reversible, post-translational modification of histones. Possible modifications are
- N-Methylation (Arg and Lys)
- N-Acetylation
- Biotinylation
- Ubiquitinylation
Formation of inactive heterochromatin is marked by methylation of a particular lysine residue (K9) on a specific histone (H3). Active chromatin on the other hand is generally associated with methylation of K4 on the same histone as well as acetylation of lysine residues in general. While the activation of chromatin via acetylation is performed by histone acetyltransferase (HAT), its deactivation is facilitated by histone deacetylases (HDACs). Study of histone acetylation is therefore closely linked to the study of these two types of enzymes.
Acetylated histones do not only play a vital part in the transcription of DNA. Their presence or absence is also associated with human diseases, such as
- Inflammatory diseases: Inhibition of HDACs leads to increased presence of acetylated histones and increased expression of inflammatory genes, after activation with inflammatory stimuli. In chronic obstructive pulmonary disease, a reduction of HDAC activity can be observed in the lung parenchyma, corresponding to the severity of the disease. Similarly, increase in HAT has been observed in bronchial biopsies from patients with asthma.
- Addiction: Chronic drug use, such as cocaine or alcohol abuse, initiates decreased HDAC activity in reward and learning-related brain regions (nucleus accumbens, prefrontal cortex or hippocampus). Acute administration of alcohol or cocaine on the other hand briefly increases the acetylation of H3, highlighting the mechanistic differences between acute and chronic covalent chemical changes. The transition between occasional drug use and its chronic counterpart is potentially caused by a single class of HDACs. HDAC5 is a deacetylase, which is phosphorylated and subsequently expelled from the nucleus upon stimulation by either drug use or stress. Through this decreased deacetylation, gene transcription and behavioral adaption can take place. HDAC5 is generally regenerated within 24 hours. However, during chronic stimulation this regeneration cannot take place, leading to an increased expression of stress- and addiction-associated genes, such as ΔFosB.
- Cancer: Abnormal alterations in histone acetylation are associated with the development of cancer. For instance, loss of acetylation at Lys16 of H4 is reported as a common abnormality in human cancer and low levels of acetylation at Lys18 of H3 was found to be a predictor of poor survival rates in pancreatic, breast, prostate and lung cancer. Thus – contrary to inflammatory diseases – inhibition of HDACs has been associated with a beneficial effect in the treatment of cancer. Several HDAC inhibitors have been approved by the FDA for the treatment of cancer
| – Vorinostat (cutaneous T-cell lymphoma) | |
| – Belinostat (peripheral T-cell lymphoma) | |
| – Panabiostat (multiple myeloma) | |
| – Romidepsin (cutaneous T-cell lymphoma) |
Due to the importance of histone acetylation for disease research, Bachem not only offers acetylated histone sequences but also HDAC substrates as part of its catalog portfolio.
Modification with larger molecules
Contrary to the modifications with small substituents (methylation, acetylation), post-translational modifications occur much more rarely with larger molecules such as biotin and ubiquitin (which can already be classified as a protein).
Nevertheless, these modifications play an important role for cellular processes – for instance, poly-ubiquitylation commonly flags proteins for degradation and mono-ubiquitylation of H2A and H2B has been associated with regulation of gene expression.
Histone biotinylation is an especially rare post-translational modification, with less than 0.001% of human histones H3 and H4 being biotinylated in nature. The biological importance of this modification is however not to be underestimated. For instance, fibroblasts of patients suffering from holocarboxylase synthetase deficiency show severely lowered rates of histone biotinylation. There is also evidence that biotinylation is part of the cellular response to DNA damage, since the global biotinylation increases upon formation of thymine dimers in lymphoid cells (caused by UV irradiation). The role of biotinylation in signaling DNA damage is further corroborated by the rapid decrease in biotinylation of Lys12 in H4 upon double-stranded DNA breaks.
Bachem offers various biotinylated histone sequences in its catalog and biotinylation is a standard modification, which can be offered as part of our custom peptide synthesis service.
References
P.J.Barnes, I.M.Adcock, K.Ito, Histone acetylation and deacetylation: importance in inflammatory lung diseases. Eur. Respir. J. 25, 552-563 (2005)
N.Kothapalli et al., Biological functions of biotinylated histones. J. Nutr. Biochem. 16, 446-448 (2005)
I.Cohen, E.Poreba, K.Kamieniarz, R.Schneider, Histone modifiers in cancer: friends or foes? Genes Cancer 2, 631-647 (2011)
T.Kuroishi, L.Rios-Avila, V.Pestinger, S.S.Wijeratne, J.Zempleni, Biotinylation is a natural, albeit rare, modification of human histones. Mol. Genet. Metab. 104, 537-545 (2011)
L.N.Hitchcock, K.M.Lattal, Histone-mediated epigenetics in addiction. Prog. Mol. Biol. Transl. Sci. 128, 51-87 (2014)
M.M.Muller, T.W.Muir, Histones: at the crossroads of peptide and protein chemistry. Chem. Rev. 115, 2296-2349 (2015)
J.L.Cadet, Epigenetics of stress, addiction, and resilience: Therapeutic implications. Mol. Neurobiol. 53, 545-560 (2016)
T.Eckschlager, J.Plch, M.Stiborova, J.Hrabeta, Histone deacetylase inhibitors as anticancer drugs. Int. J. Mol. Sci. 18, (2017)
DEVELOPMENT OF PEPTIDE HISTONE DEACETYLASE INHIBITORS AS DRUGS
Unbalanced histone acetyltransferase (HAT) and histone deacetylase (HDAC) enzyme activity is involved in the development and progression of many different types of cancers. HDAC inhibitors increase the levels of acetylated lysine residues in histones, restarting the expression of silenced regulatory genes in cancerous cells. HDAC inhibitors induce cell cycle arrest, apoptosis, reduce angiogenesis and modulate immune response. Therefore, HDAC inhibitors are of interest for the treatment of cancer. So far, the U.S. Food and Drug Administration (FDA) has approved four HDAC inhibitors. One of these drugs is a bicyclic depsipeptide known as Istodax® (romidepsin) which the FDA approved in 2009 for the treatment of cutaneous T-cell lymphoma (1). In addition to Istodax, there are other peptidic HDAC inhibitors in clinical and preclinical development as shown in Table 1.
| Product Name | Active Ingredient | Condition Treated | Highest Phase | Company |
|---|---|---|---|---|
| OBP801 | spiruchostatin A | Solid Tumor, Glaucoma | Phase I | Oncolys Biopharma Inc |
| OKI-179 | -- | Breast Cancer | Preclinical | OnKure Inc |
| Chromopeptide A | -- | Prostate Cancer | Preclinical | Zhejiang Hisun Pharmaceutical Co Ltd |
Phase I
Oncolys Biopharma is developing OBP801 (spiruchostatin A), a cyclic depsipeptide, for the treatment of various solid cancers and glaucoma. OBP801 acts by targeting histone deacetylase. In 2015, Oncolys Biopharma initiated a Phase Ia study of OBP801 in patients with advanced solid tumors (3). In addition, OBP801 is in preclinical development for glaucoma. In 2016, Oncolys BioPharma entered into a collaboration with Kyoto Prefectural University of Medicine to research the potential application of OBP801 for the prevention of post-surgery scarring in glaucoma patients (2).
Preclinical Candidates
OnKure Inc, a University of Colorado spinoff, is developing OKI-179 for the treatment of triple-negative breast cancer (2). OKI-179 is a derivative of largazole, a cyclic depsipeptide that was isolated from the marine cyanobacterium Symploca sp. (4). This drug candidate targets HDAC6. In 2018, OnKure raised USD 7 million Series A financing. The company plans to use the funds for the development of OKI-179 and other drug candidates in its pipeline (2).
Chromopeptide A is under development by Zhejiang Hisun Pharmaceutical for the treatment of prostate cancer. This drug candidate is a depsipeptide originally isolated from the marine sediment-derived bacterium Chromobacterium sp. Chromopeptide A targets HDAC1, HDAC2, HDAC3 and HDAC8 to stop uncontrolled proliferation and differentiation of cancer cells. In preclinical trials, Zhejiang Hisun Pharmaceutical found that Chromopeptide A suppressed the growth and migration of prostate cancer in vitro and in vivo (2).
Conclusion
HDAC inhibitors are emerging as anti-cancer drugs and they may have potential beyond cancer. To support researchers and organizations who are studying histones and histone deacetylases, Bachem offers a selection of histone fragments, histone deacetylase substrates and histone building blocks. In addition, Bachem offers a comprehensive custom peptide synthesis service and the production of New Chemical Entities.
References
(1) A.K. Singh et al., Targeting histone deacetylases with natural and synthetic agents: An emerging anticancer strategy, Nutrients. 10(6), 731 (2018)
(2) GlobalData (2018)
(3) Medtrack (2018)
(4) K. Taori et al., Structure and activity of largazole, a potent antiproliferative agent from the Floridian marine cyanobacterium Symploca sp., J. Am. Chem. Soc. 130(6), 1806-1807 (2018)
MEET BACHEM: IAIN PRITCHARD
What is your official job title at Bachem?
I am the Director of Production at Bachem UK.
How long have you been with Bachem? Where did you work before Bachem?
I have worked for Bachem for almost 15 years now. I joined the company as a Synthesis Chemist not long after completing my undergraduate studies. I became Synthesis Manager a few years later before assuming my current role in 2013.
What is your academic background/degrees or training?
I have a BSc in Forensic & Analytical Chemistry and then whilst working with Bachem, I completed my PhD in Investigative Medicine.
What do you like to do outside of work?
Spending time with family, friends and at the Liverpool Football Club.
What is your business motto?
When I was younger my father always told me “if a job is worth doing, then it is worth doing properly”. I still find the relevance in that statement every single day.
What do you like most about your job?
The variety. At Bachem UK we work on a vast number of different projects at any one time and I am fortunate enough to be involved in the majority of them. As a result, every day brings different challenges.
What is your preferred peptide?
I’d have to say Peptide YY as it formed the basis of my PhD studies. Even now, many years later I could probably still recount the amino acid sequence without even checking!
Thank you very much Iain.

Peptide highlights
Interesting news about peptides in basic research and pharmaceutical development:
Promising new therapy spares muscle loss in Duchenne muscular dystrophy-University at Buffalo
New research reveals a mitochondrial gene that protects against dementia and other diseases of aging-University of Southern California
New hurdle for developing immunotherapies-Science Daily
Cyclic peptides for cancer therapy and prognosis assessment-Medical Xpress
LITERATURE CITATIONS
Bachem peptides and biochemicals are widely cited in research publications. Congratulations to all our customers with recent publications!
R.E. Bonomi et al.
A novel substrate radiotracer for molecular imaging of SIRT2 expression and activity with positron emission tomography.
Molecular imaging and biology 20, 594-604 (2018)
L.C. Chen et al.
Design of diarylheptanoid derivatives as dual inhibitors against class IIa histone deacetylase and beta-amyloid aggregation.
Frontiers in Pharmacology 9, 708 (2018)
T. Nakatani et al.
The deletion of Hdac4 in mouse osteoblasts influences both catabolic and anabolic effects in bone.
Journal of Bone and Mineral Research 33, 1362-1375 (2018)
D.S. Reddy et al.
Measuring histone deacetylase inhibition in the brain.
