MEET US AT BOULDER PEPTIDE SYMPOSIUM

Boulder Peptide Symposium (BPS) 2019 will take place on September 23 – 26, 2019 at the St Julien Hotel and Spa, in Boulder, CO.
The Boulder Peptide Society, the organizer of the BPS, is a non-profit organization dedicated to supporting the advancement of peptide therapeutics. This annual meeting brings together key decision leaders from industry and academia to discuss and address the challenges in the development, delivery and stability of peptide therapeutics.
Bachem provides a full range of services to the pharma and biotech industries. It specializes in the development of innovative, efficient manufacturing processes and the reliable production of peptide-based active pharmaceutical ingredients. Bachem‘s pipeline contains more than 150 customer projects in preclinical and clinical phases with promising potential: in the last couple of years, a number of products in phase III trials received marketing authorization and phase II projects progressed to phase III clinical trials.
Join the Bachem presentation on Wednesday, September 25, 1:30 – 2:00 pm: Daniel Samson, Vice President API SPPS at Bachem AG, will present “CMC Development Concept for Synthetic Peptides”.
The Bachem team is excited to meet with you, learn your needs for peptides and discuss how Bachem can meet your API custom manufacturing needs. We kindly invite you to drop by our Booth #4 or to contact us to schedule a meeting in advance.
We look forward to meeting you at Boulder Peptide Symposium 2019!
MELANOCORTIN PEPTIDES AND THEIR RECEPTORS
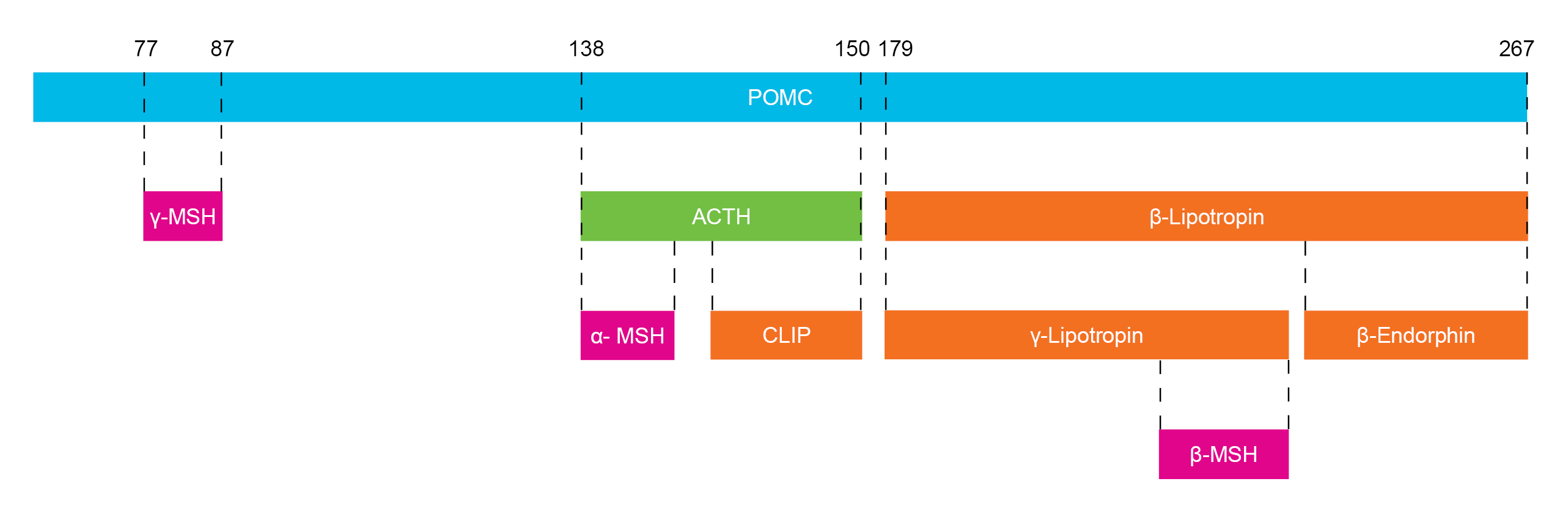
Melanocortins are a family of peptides comprising ACTH (adrenocorticotropic hormone) and the melanocyte stimulating hormones α-MSH, β-MSH and γ-MSH. They are derived from the common precursor POMC (pro-opiomelancortin) (Figure 1) and act via the melanocortin receptors (MC1R-MC5R). Melanocortins are produced via a series of enzymatic steps in a tissue-specific manner and are involved in the regulation of a diverse range of physiological functions including pigmentation, steroidogenesis, energy homeostasis and eating behavior.
Figure 1 Melanocortin Peptides Derived from POMC (human)
(ACTH: Adrenocorticotropic Hormone, CLIP: Corticotropin-Like Intermediary Peptide, MSH: Melanocyte Stimulating Hormone, POMC: Pro-opiomelanocortin)
Melanocortin Peptides
Adrenocorticotropic Hormone (ACTH)
ACTH is a 39 amino acid peptide, which is produced in cells of the anterior pituitary under the influence of the hypothalamic hormones corticotropin-releasing hormone (CRH) and Arg8-vasopressin (AVP). The primary role of ACTH is regulating the synthesis and release of glucocorticoids by the adrenal gland. In humans, cortisol (hydrocortisone) is the principle circulating glucocorticoid. ACTH acts via MC2R in the zona fasciculata of the adrenal cortex. MC2R expression and function is dependent on the MC2R accessory protein, MRAP1, which is a single-pass transmembrane protein highly expressed in the adrenal gland.
The physiological effect of glucocorticoids consists primarily in stimulating gluconeogenesis from amino acids and inhibiting glucose oxidation in the cells and the associated increase in blood sugar levels. Since the amino acids used for gluconeogenesis originate from increased protein degradation in the peripheral organs (muscle tissue, bone matrix), glucocorticoids have a catabolic effect.
Melanocyte Stimulating Hormones (α-MSH, β-MSH, γ-MSH)
α-MSH, β-MSH, and γ-MSH are derived from different regions of POMC. In mammals, mature α-MSH consists of 13 amino acids and can be produced by the further processing of ACTH including formation of a C-terminal amide and N-terminal acetylation. α-MSH is expressed in the hypothalamic arcuate nucleus and in the nucleus tractus solitarius of the brainstem where it has a crucial role in the regulation of metabolic functions. When administered centrally, α-MSH suppresses feeding in free-feeding or fasted rodents.
α-MSH is also found in the skin. Epidermal cells such as keratinocytes and melanocytes synthesize and release α-MSH after stimulation with pro-inflammatory cytokines or UV light. α-MSH acts via MC1R and triggers the switch from red/yellow pheomelanin to brown/black eumelanin production. In addition to its pigment inducing capacity, α-MSH is able to modify keratinocyte proliferation and differentiation.
β-MSH is generated by processing of β-lipotropin which also yields γ-lipotropin and β-endorphin. The existence of β-MSH is controversial. It has been suggested that human β-MSH does not exist in nature, but is an artifactually produced fragment of the β-lipotropin molecule. A recent study, however, suggests that β-MSH is a true neuropeptide and that the predominant species of β-MSH is the 18-amino-acid form predicted by the location of dibasic cleavage sites.
γ-MSH is encoded in the N-terminal region of POMC and exists in three forms γ1-MSH, γ2-MSH, and γ3-MSH. γ-MSH is an agonist of MC1R, MC3R, MC4R, and MC5R. As γ-MSH has the highest affinity for MC3R it is considered to be the natural ligand of this receptor.
Melanocortin Receptors (MC1R – MC5R)
The five melanocortin receptors MC1R-MC5R belong to the family of class A (rhodopsin-like) G protein-coupled receptors. They exhibit differential tissue expression and functions and are functionally coupled to adenyl cyclase. Table 1 illustrates the agonist affinities and main functions of MCRs. MCRs mediate their effects primarily by activating a cAMP-dependent signaling pathway. In addition to melanocortin peptides, other endogenous peptides such as the agouti signaling protein (ASIP), agouti-related protein (AgRP), and β-defensin 3 can bind to these receptors. ASIP is a 131 amino acid protein produced in the hair follicle. It acts on follicular melanocytes to inhibit α-MSH induced eumelanin production. Pharmacological studies indicate that ASIP is a selective antagonist for MC1R and MC4R.
AgRP consists of 112 amino acids and is a potent, selective antagonist of MC3R and MC4R. AgRP is expressed in the adrenal cortex and in the hypothalamus of the brain where it antagonizes the action of α-MSH. Orexigenic AgRP neurons in the arcuate nucleus (Arc) of the hypothalamus are critical regulators of feeding and food-seeking behavior.
One of the major β-defensins produced in skin, β-defensin 3, has been discovered to function as an MCR ligand in vivo and in vitro. Human β-defensin 3 acts as a neutral MCR antagonist capable of blocking the action of either stimulatory agonists such as α-MSH or inhibitory inverse agonists such as ASIP and AgRP.
| MCR | Agonist Affinity | Main Function | |
|---|---|---|---|
| MC1R | α-MSH ≥ ACTH = β-MSH > γ-MSH | Pigmentation | |
| MC2R | ACTH | Adrenal steroidogenesis | |
| MC3R | γ-MSH = α-MSH = ACTH > β-MSH | Energy rheostasis | |
| MC4R | α-MSH ≥ ACTH > β-MSH > γ-MSH | Energy homeostasis | |
| MC5R | α-MSH ≥ ACTH > β-MSH > γ-MSH | Exocrine gland secretion |
Adapted from A.R.Rodrigues, H.Almeida, A.M.Gouveia, Intracellular signaling mechanisms of the melanocortin receptors: current state of the art. Cellular and Molecular Life Sciences 2015, 72, 1331-1345.
MC1R
MC1R is primarily expressed in melanocytes of the skin and is involved in skin pigmentation and DNA damage response, including DNA repair pathways. ACTH and α-MSH are considered the physiological agonists of MC1R.
Human melanocytes can produce two types of melanins, the red/yellow pheomelanin and the brown/black eumelanin. They are synthesized within specific organelles, the melanosomes, regardless of the pigmentary phenotype of the skin. For example, individuals with red hair have a predominance of pheomelanin in hair and skin and/or a reduced ability to produce eumelanin.
UV-induced pigmentation requires induction of α-MSH secretion by keratinocytes and is triggered by DNA damage and subsequent transcriptional activation of POMC by the tumor-suppressor protein p53. Elevated expression of the POMC protein and production of α-MSH results in MC1R activation and increased synthesis of eumelanin, which is highly photoprotective by absorbing UV radiation.
Certain variations (polymorphisms) in the MC1R gene are associated with red hair color, fair skin, and increased cancer risk.
MC2R
MC2R is mainly expressed in the adrenal glands. Activation of MC2R by ACTH mediates the signaling cascade leading to steroid synthesis.
25% of familial glucocorticoid deficiency (FGD) are caused by mutations in MC2R. FGD is a rare, early onset disorder characterized by glucocorticoid deficiency, higher plasma ACTH and normal mineralocorticoid levels. Elevated ACTH levels can lead to hyperpigmentation due to activation of MC1R.
MC3R
MC3R is primarily found in the central nervous system especially in the hypothalamus but is also expressed in the periphery such as the gut and kidney. It can be activated by α-, β-, and γ-MSH as well as by ACTH. γ-MSH exhibits a 100-fold higher affinity and 45-fold higher potency at the MC3R than at the other MCRs.
MC3R appears to be involved in metabolic regulation. A recent study points at a role of MC3R in the regulation of energy rheostasis. The findings indicate that MC3R is required bidirectionally for controlling responses to external homeostatic challenges, such as caloric restriction or calorie-rich diet. Mice deficient in MC3R lack this control and lost more weight when fasting and gained more weight when eating a high-fat diet, compared with normal mice.
MC4R
As MC3R, MC4R is also expressed in the central nervous system. In adult rat brain, it can be found in several regions including cortex, thalamus, hypothalamus, and brainstem. In the hypothalamus, it is expressed highly in the paraventricular nucleus (PVN).
MC4R is an essential regulator of energy homeostasis and metabolism and has high affinity for α-MSH and ACTH. Stimulation of MC4R decreases food intake and increases energy expenditure. Loss of function mutations of MC4R are associated with obesity. It has been suggested that defects in the MC4R gene are responsible for 2%-6% of all cases.
MC5R
MC5R is highly expressed in multiple exocrine tissues and might play a role in the regulation of a variety of exocrine glands. Targeted disruption of the MC5R produced mice with a severe defect in water repulsion and thermoregulation due to decreased production of sebaceous lipids. MC5R has also been suggested to be involved in pheromone-regulated behavior in mice.
Summary
The melanocortin system is involved in a wide variety of physiological functions such as skin pigmentation, steroidogenesis, energy homeostasis and metabolism. Therefore, it represents an important therapeutic target for various disorders including obesity, cachexia and inflammatory diseases. In order to support your research in these areas, Bachem offers a range of well characterized MCR agonists and antagonists (see Table 2).
For more information please see our online shop and our products related to obesity and inflammation research.
| Product Number | Product Name | Synonym | Agonist/Antagonist |
|---|---|---|---|
| 4008476 | α-MSH | α-Melanotropin | Agonist |
| 4030885 | β-MSH (human) | Agonist | |
| 4008707 | γ₁-MSH | Agonist | |
| 4008709 | γ₂-MSH | Agonist | |
| 4030911 | γ₃-MSH | Agonist | |
| 4003800 | (Nle⁴,D-Phe⁷)-α-MSH | Afamelanotide | Agonist |
| 4039778 | Acetyl-(Nle⁴,Asp⁵,D-Phe⁷,Lys¹⁰)-cyclo-α-MSH (4-10) amide | Melanotan II | Agonist |
| 4028679 | Acetyl-(Cys¹¹,D-2-Nal¹⁴,Cys¹⁸)-β-MSH (11-22) amide | HS014 | Antagonist |
| 4029134 | Acetyl-(Cys³,Nle⁴,Arg⁵,D-2-Nal⁷,Cys¹¹)-α-MSH (3-11) amide | HS024 | Antagonist |
| 4034459 | (Deamino-Cys¹¹,D-2-Nal¹⁴,Cys¹⁸)-β-MSH (11-22) amide | JKC-363 | Antagonist |
| 4034460 | (Deamino-Cys³,Nle⁴,Arg⁵,D-2-Nal⁷,Cys¹¹)-α-MSH (3-11) amide | JKC-366 | Antagonist |
| 4026419 | (Met⁵,Pro⁶,D-Phe⁷,D-Trp⁹,Phe¹⁰)-α-MSH (5-13) | Nonapeptide-1 | Antagonist |
| 4027601 | Acetyl-(Nle⁴,Asp⁵,D-2-Nal⁷,Lys¹⁰)-cyclo-α-MSH (4-10) amide | SHU9119 | Antagonist |
References
M.Ghamari-Langroudi, I.Cakir, R.N.Lippert, P.Sweeney, M.J.Litt, K.L.J.Ellacott et al, Regulation of energy rheostasis by the melanocortin-3 receptor. Science Advances 2018, 4, eaat0866.
P.Kirwan, R.G.Kay, B.Brouwers, V.Herranz-Perez, M.Jura, P.Larraufie et al, Quantitative mass spectrometry for human melanocortin peptides in vitro and in vivo suggests prominent roles for beta-MSH and desacetyl alpha-MSH in energy homeostasis. Molecular Metabolism 2018, 17, 82-97.
T.V.Novoselova, L.F.Chan, A.J.L.Clark, Pathophysiology of melanocortin receptors and their accessory proteins. Best Practice & Research Clinical Endocrinology & Metabolism 2018, 32, 93-106.
Y.Yang, C.M.Harmon, Molecular signatures of human melanocortin receptors for ligand binding and signaling. Biochimica et Biophysica Acta. Molecular Basis of Disease 2017, 1863, 2436-2447.
A.R.Rodrigues, H.Almeida, A.M.Gouveia, Intracellular signaling mechanisms of the melanocortin receptors: current state of the art. Cellular and Molecular Life Sciences 2015, 72, 1331-1345.
J.C.Garcia-Borron, Z.Abdel-Malek, C.Jimenez-Cervantes, MC1R, the cAMP pathway, and the response to solar UV: extending the horizon beyond pigmentation. Pigment Cell and Melanoma Research 2014, 27, 699-720.
M.A.Nix, C.B.Kaelin, T.Ta, A.Weis, G.J.Morton, G.S.Barsh et al, Molecular and functional analysis of human beta-defensin 3 action at melanocortin receptors. Chemistry and Biology 2013, 20, 784-795.
S.Ramachandrappa, R.J. Gorrigan, A.J. Clark, L.F. Chan, The melanocortin receptors and their accessory proteins. Frontiers in Endocrinology (Lausanne) 2013, 4, 9.
B.J.Renquist, R.N.Lippert, J.A.Sebag, K.L.Ellacott, R.D.Cone, Physiological roles of the melanocortin MC(3) receptor. European Journal of Pharmacology 2011, 660, 13-20.
Y.X.Tao, The melanocortin-4 receptor: physiology, pharmacology, and pathophysiology. Endocrine Reviews 2010, 31, 506-543.
G.W.Millington, The role of proopiomelanocortin (POMC) neurones in feeding behaviour. Nutrition & Metabolism 2007, 4, 18.
PEPTIDES IN DEVELOPMENT TARGETING MELANOCORTIN RECEPTORS
The melanocortin receptor system is involved in important physiological functions such as feeding behavior, response to UV radiation, sexual function, pain response and more. Accordingly, melanocortin receptors are involved in diseases such as obesity, sexual dysfunction, cancer and skin disorders (1). There are already approved drugs targeting melanocortin receptors such as corticotropin (Acthar®), cosyntropin (Synacthen®), afamelanotide (Scenesse®), and bremelanotide (VylessiTM) which was approved by the U.S. Food and Drug Administration (FDA) in June 2019 for the treatment of hypoactive sexual desire disorder in premenopausal women. There is ongoing interest in the melanocortin receptor system and additional drug candidates targeting melanocortin receptors are in clinical development as shown in Table 1.
| Product Name | Target | Pipeline Indication(s) | Highest Development Stage |
|---|---|---|---|
| Setmelanotide | MC4R | POMC deficiency obesity, LEPR deficiency obesity, Bardet-Biedel syndrome, Alström syndrome, POMC Heterozygous deficient obesity, POMC epigenetic disorders | III |
| PL-8177 | MC1R | Uveitis, Ulcerative Colitis, Diabetic Retinopathy, Dermatological Disorders, Kidney Disease (Nephropathy) | I |
Setmelanotide, an MC4R agonist, is under development by Rhythm Pharmaceuticals for the treatment of rare genetic obesity disorders such as POMC deficiency obesity, leptin receptor-deficiency (LEPR) obesity, Bardet-Biedl syndrome and Alström syndrome. MC4R is part of a biological pathway that regulates weight by increasing energy expenditure and reducing appetite. By activating MC4R, setmelanotide therapy results in a decrease in food intake (2). In 2019, Rhythm announced promising interim clinical data in MC4R pathway heterozygous (HET) obesity. The preliminary data show consistent weight loss and hunger decreases in patients with high-impact loss-of-function variants (3).
Rhythm Pharmaceuticals is also developing a once-weekly extended release formulation of setmelanotide. In 2016, Rhythm Pharmaceuticals and Camurus entered into a license agreement for Camurus’ drug delivery technology known as FluidCyrstal® to be applied in the development of a long acting formulation of setmelanotide (2).
Palatin Technologies is developing PL-8177, an MC1R agonist, for the treatment of ulcerative colitis and other inflammatory bowel diseases. It is also in the preclinical stage of development for diabetic retinopathy and nephritis. PL-8177 is a synthetic cyclic heptapeptide and a potent agonist of MC1R. In 2018, Palatin announced positive top line results from a clinical study of PL-8177 using an oral delayed release polymer formulation of PL-8177. In 2019, the FDA granted orphan drug designation for PL-8177 for the treatment of non-infectious intermediate, posterior, pan and chronic anterior uveitis (2).
Conclusion
Due to the multifaceted roles of melanocortin receptors, targeting these receptors holds promise for drug discovery across multiple therapeutic areas. For researchers and organizations studying the melanocortin receptor system, Bachem offers a selection of research products including ACTH sequences and Melanocyte-Stimulating Hormone (MSH) sequences. In addition, Bachem offers a comprehensive custom peptide synthesis service, a selection of Generic Active Pharmaceutical Ingredientss including Tetracosactide (cosyntropin) and the production of New Chemical Entities.
References
(1) M.Cai and V.J.Hruby, The melanocortin receptor system: A target for multiple degenerative diseases. Curr. Protein Pept. Sci, 17(5), 488-496 (2019)
(2) GlobalData (2019)
(3) Rhythm Pharmaceuticals announces promising clinical data in MC4R pathway heterozygous obesity and strategy for further development, Rhythm (2019)
MEET BACHEM: DENNIS RANZ
What is your official job title at Bachem?
I work as Chemical Laboratory Technician in Quality Control.
How long have you been with Bachem? Where did you work before Bachem?
I started working at Bachem in July 2015. In my first position at Bachem, I have worked in Sampling and after that I changed to the wet laboratory of GMP release. Before joining Bachem, I worked for 3 years in the operating laboratory of an international company.
Briefly, what do you do at Bachem?
I am responsible for the release analysis. This contains the analysis of key and non-key intermediates, IPC studies, stress tests, validations, R&D and customer samples.
What is your academic background or training?
I have an education as a Chemical Technical Assistant. After my apprenticeship, I completed a training as Bachelor Professional of Chemical Production and Management at IHK.
What do you like to do outside of work?
I love the nature. Therefore I am fond of mountain biking, paragliding and hiking. In addition, I like modelling airplanes with my dad. I play squash and also play the clarinet in three orchestras.
What makes a perfect day for you?
Standing up healthy, spending my time outside, especially in the mountains and having my beloved ones around me.
Thank you very much Dennis.

Peptide highlights
Interesting news about peptides in basic research and pharmaceutical development:
‘Limitless potential’ of artificial protein ushers in new era of ‘smart’ cell therapies-University of California San Francisco
Expanding functions of conducting microbial nanowires for chemical, biological sensors-Science Daily
New tuberculosis tests pave way for cow vaccination programs-Penn State
How peptides arose from the primordial soup-Chemical & Engineering News
LITERATURE CITATIONS
Bachem peptides and biochemicals are widely cited in research publications. Congratulations to all our customers with recent publications!
M.Chen et al.
Gsα deficiency in the dorsomedial hypothalamus leads to obesity, hyperphagia, and reduced thermogenesis associated with impaired leptin signaling.
Molecular Metabolism 25, 142-153 (2019)
N.Lucas et al.
Immunoglobulin G modulation of the melanocortin 4 receptor signaling in obesity and eating disorders.
Translational Psychiatry 9, 87 (2019)
D.Ramírez et al.
NDP-MSH reduces oxidative damage induced by palmitic acid in primary astrocytes.
Journal of Neuroendocrinology 31, e12673 (2019)
F.Saporiti et al.
Melanocortin-1 receptor positively regulates human artery endothelial cell migration.
