PEGYLATION OF PEPTIDES
Bachem offers PEGylation as a cost-effective modification of peptides which has the potential to improve bioavailability compared to the unmodified molecule.
Active pharmaceutical ingredients (APIs) based on synthetic peptides are recognized as potent and selective drugs. Endogenous peptides such as peptide hormones are involved in complex biological processes in a specific and precise manner. Many of these peptides have been sequenced and thus can be synthetically manufactured. Other pharmaceutically active peptides have been rationally designed, either by modifying or optimizing a native sequence or by de novo screening methods to produce peptide mimetics with little or no homology to the native protein.
However, since 1970 only about 30 of these highly potent compounds have been synthetically manufactured at large scale for approved pharmaceutical applications. One reason for the low market share of peptides in the pharmaceutical market is the high cost of administration in therapeutic settings. This unfortunate reputation persists even as increased scales of manufacturing drive down the cost of raw materials and the recent advances in peptide manufacturing and scale-up protocols improve manufacturing efficiencies. For any given clinical utility of a drug, reducing cost-of-goods at the point of use and increasing bioavailability in vivo are major factors in determining the drug’s cost and success or failure in the marketplace.
Limitations by the peptide
The in vivo bioavailability of a typical peptide drug is limited by its low circulating half-life which could measure few hours or, even more likely, a few minutes. This is due to enzymatic digestion and other endogenous factors, such as renal clearance. Due to their low toxicity relative to many other classes of drugs, the short half-life of peptides can often be compensated for by a higher than therapeutic level of dosing. However, the administration of peptide drugs by high-dose injection can lead to wide fluctuations in blood levels that can be detrimental to a sustained therapeutic response. This problem is therefore usually mitigated by more frequent dosing of the peptide. In the treatment of chronic diseases in particular, more frequent dosing of peptide drugs translates into reduced patient compliance, higher costs and a greater chance for adverse events and side effects.
Peptides can cost many thousands of dollars per gram to manufacture. The costs of synthesis increase with the number of amino acids and can be reduced per unit by upscaling.
At an equivalent level of purity, a 36 amino acid peptide with over 100 manufacturing steps can be 10-20 times more expensive than a pentapeptide synthesized in 6 steps. Add to this the cost of formulating, packaging, distribution etc. and the cost per dose increases significantly. Used in chronic treatment protocols as an injectable, a highly potent peptide with a typically short half-life often requires frequent dosing. This can cost thousands of dollars/patient/year.
During the past decades, pharmaceutical companies concentrated on the development of small molecules and left peptide development for large biotech companies. However, biotech cash life is often measured in months instead of years and proof-of-concept study data from human subjects remains the fastest avenue to investor and partnership funding and company longevity. Therefore, these companies have traditionally relied on evidence of clinical efficacy from an injection protocol, using a peptide sequence that may have been modified but which remains essentially “naked”. Sometimes stabilizers, excipients and bulking agents are added to contribute to the stability of the naked peptide in a solution dose form. More often, peptides are stored in powder form, as a lyophilizate, ready to be dissolved at the point of use.
Modification and performance
Chemical modification of the peptide using polyethylene glycol (PEG) can improve drug performance with minimal increase in manufacturing cost. PEG is a highly investigated polymer that is used in covalent modification of biopolymers as proteins and peptides. It is incorporated into the manufacturing process of the bulk API in a technique known as PEGylation. The effects of PEGylation on peptide pharmacokinetics include avoidance of reticuloendothelial (RES) clearance, mitigation of immunogenicity, and reduction of enzymatic proteolysis and of losses by renal filtration, with potentially beneficial changes in biodistribution. These effects can dramatically increase the half-life of a peptide in vivo, with potential collateral improvement in bioavailability but without adversely affecting binding and activity of the peptide ligand.
PEG’s most common form is a linear or branched polyether with terminal hydroxyl groups synthesized by anionic ring opening polymerization – HO-(CH2CH2O)n-CH2CH2-OH (Fig. 1).
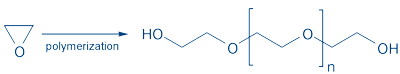
Figure 1: Polymerization of ethylene oxide.
Monofunctional methoxy-PEG (mPEG) is preferred for peptide modification – CH3O-(CH2CH2O)n-CH2CH2-OH, as it can be derivatized with a number of linkage moieties, yielding methoxy-PEG-amines, -maleimides, or -carboxylic acids (Fig. 2).
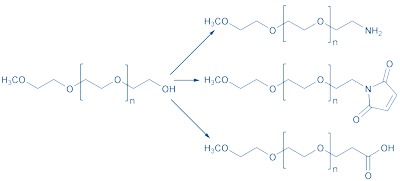
Figure 2: Modifications of mono-methylated polyethylene glycol (mPEG).
Four general factors affect the performance of PEGylated peptides:
1. Molecular weight and structure – whereas PEGs of <1,000 Da can be broken down into subunits that can have some toxicity, PEGs of >1,000 Da have not demonstrated any toxicity in vivo. PEGs of up to 40-50,000 Da have been used in clinical and approved pharmaceutical applications.
2. Number of PEG chains – two or more lower-weight chains can be added to increase the total molecular weight of the PEG complex.
3. Site of attachment – for each peptide, the location of the PEGylation sites has to be carefully engineered experimentally to retain the highest possible binding efficiency and activity of the peptide ligand.
4. PEGylation chemistry – the type of linkage for attaching PEG to the peptide as well as the purity of raw materials, intermediates and final product.
The latter is the most important factor determining the yield of the PEGylation process and the scalability of the manufacturing protocol. Peptide and linker have to be very pure and very stable during the conjugation reaction to yield a pure conjugate with high efficiency.
PEGylation chemistry
The first step in coupling PEG monomethyl ether to a peptide is to activate mPEG with a functional group. Its nature depends on the available reactive groups on the peptide, such as lysine, aspartic acid, cysteine, glutamic acid, serine, threonine, the N-terminal amine and the C-terminal carboxylic acid or other specific sites (Fig. 3).
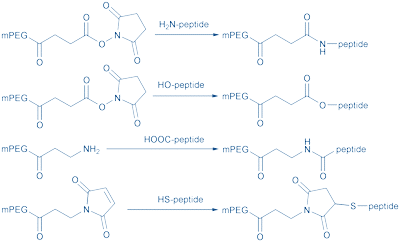
Figure 3: Conjugation of peptides to mPEG.
Both weak and relatively strong linkages to the peptide can be used, although strong linkage is consistent with the objective of increasing the circulating half-life of the peptide ligand and its stability in solution. A weak PEG-peptide linkage is desired in peptide prodrugs. For example, the imidazole of a histidine residue is reacted with mPEG succinimidyl carbonate under slightly acidic conditions. The resulting base-labile peptide-PEG carbamate linkage is hydrolyzed in a controlled fashion in vivo. This slow release can be chemically fine-tuned to occur in endogenous circulation over hours, days or weeks.
Examples of PEG chemistry include modifications of free cysteine. These are possible with selective reagents, if sufficient care is taken to prevent dimerization during the coupling. A variety of PEG derivatives, such as PEG-maleimide, are readily available. The conjugation of a peptide to many of these derivatives can be rate- and site-controlled by monitoring and adjusting the pH during the coupling. Carbohydrates can also be oxidized and conjugated to hydrazide PEG derivatives, but glycopeptides are not very common, and a discussion on them would exceed the scope of this article.
Finally, there is reversible PEGylation. Although the effect has not been demonstrated with peptides, some protein PEG conjugates show lower activity as a conjugate but -due to pharmacokinetics- a higher relative in vivo activity. For instance, PEGIntron (PEGylated interferon alpha-2b) has a degradable linkage which improves its half-life. PEG is coupled to the imidazole ring of histidine. The carbamate linkage is stable at pH 5. In vivo, at physiologic pH, PEG is slowly released from the protein with concomitant increase of the protein’s biological activity. Apart from linear single-arm PEG, there are various more complex PEG structures. Branched PEG is generated by attaching two linear PEGs to the alpha and epsilon groups of a lysine core, yielding a well-defined PEG containing a reactive carboxyl group (Fig. 4).
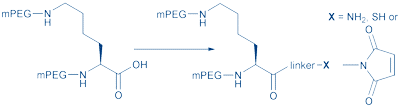
Figure 4: Bis-mPEGylated lysine derivatives for conjugation.
During synthesis, a few by-products are generated which can be removed by careful reverse-phase HPLC or ion chromatography. The lysine-based linker can then be converted into a wide range of derivatives, such as thiols and maleimides. Due to the adhered water molecules (PEG can bind 2-3 water molecules per ethylene oxide unit), the dimer PEG behaves like a much larger monomer PEG in vitro, with the biological effect of reducing renal clearance. In heterobifunctional PEG, different reactive groups are attached to the ends of a single (or dimer) PEG chain (Fig. 5).
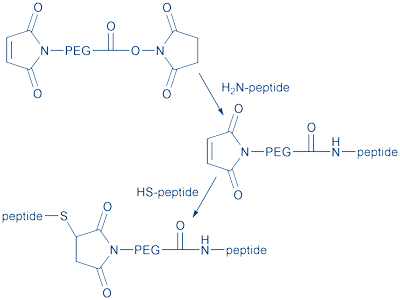
Figure 5: Heterobifunctional PEG for conjugating two different peptides.
So, the conjugates contain two peptide chains in close proximity, which may improve affinity, for instance, with dimerized cell surface receptors. It has been documented elsewhere that PEGylation with mPEG2-NHS 40 Kd decreases clearance of certain proteins, for instance interferon alfa (marketed by Roche as Pegasys®) from 9 hours for the native molecule to 77 hours for the conjugate. In peptide conjugates, DAC (Drug Affinity Construct) technology by ConjuChem Inc. uses site-specific conjugation of peptides to albumin to retain a therapeutic potency and duration of activity in circulation comparable to native albumin. This construct increases the circulating half-life of the peptide from hours to weeks.
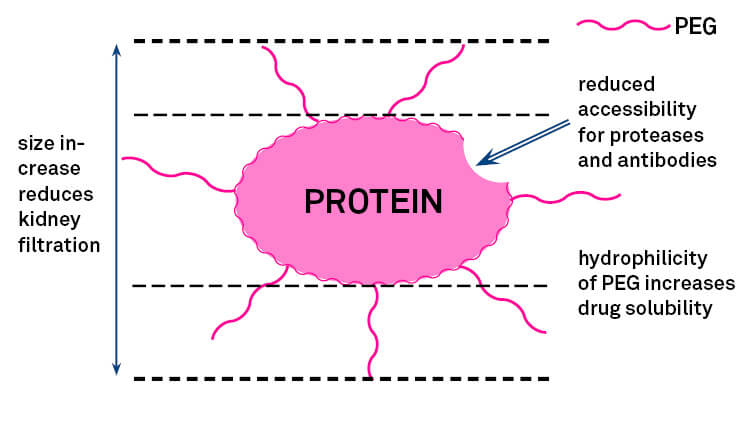
Figure 6: Advantages of PEGylation.
References
J.M. Harris and R.B. Chess, Effect of pegylation on pharmaceuticals, Nat. Rev. Drug Discov. 2, 214-221 (2003)
F.M. Veronese and G. Pasut, PEGylation, successful approach to drug delivery, Drug Discov. Today 10, 1451-1458 (2005)
F.M. Veronese and A. Mero, The impact of PEGylation on biological therapies, BioDrugs 22, 315-329 (2008)
J.S. Kang et al., Emerging PEGylated drugs, Expert Opin. Emerg. Drugs 14, 363-380 (2009)
R.W. Payne et al., Product development issues for PEGylated proteins, Pharm. Dev. Technol. 16, 423-440 (2011)
S.S. Banerjee et al., Poly(ethylene glycol)-prodrug conjugates: Concept, design, and applications, J. Drug Deliv. 103973 (2012)
P. Ingallinella et al., PEGylation of Neuromedin U yields a promising candidate for the treatment of obesity and diabetes, Bioorg. Med. Chem. 20, 4751-4759 (2012)
M. González and S.E. Vaillard, Evolution of reactive mPEG polymers for the conjugation of peptides and proteins Curr. Org. Chem. 17, 975-998 (2013)
Y. Ikeda et al., Novel protein PEGylation chemistry via glutal-aldehyde-functionalized PEG, Bioconjug. Chem. 24, 1824-1827 (2013)
P. Muralidharan et al., Inhalable PEGylated phospholipid nanocarriers and PEGylated therapeutics for respiratory delivery as aerosolized colloidal dispersions and dry powder inhalers, Pharmaceutics 6, 333-353 (2014)
J.K. Dozier and M.D. Distefano, Site-specific PEGylation of therapeutic proteins, Int. J. Mol. Sci.16, 25831-25864 (2015)
Image: PDB ID 1ITF: INTERFERON alpha-2A
Klaus, W., Gsell, B., Labhardt, A.M., Wipf, B. and Senn, H. (1997) The three-dimensional high resolution structure of human interferon alpha-2a determined by heteronuclear NMR spectroscopy in solution. J.Mol.Biol. 274: 661-675 (PubMed ID 9417943)
RARE DISEASES

Because a rare disease by definition impacts very small populations worldwide, those that suffer from these diseases have particularly intimate relationships both among one another, and with those companies pursuing treatments. Their advocacy groups are fierce and vocal with the FDA, and their loyalty runs deep for companies they view as having integrity and bravery for dedicating resources toward drugs comprising a marketplace far less established than those geared toward “traditional” indications. The sponsors of rare disease drugs seem to embody this same passion, and when it comes to manufacturing their peptides experience has shown us that they are far more hands on than the typical customer, paying excessively careful attention to daily operations, costs, timelines, etc. They are challenging customers to manage, but playing a major role in treating a rare disease is worth the extra effort.
In Paroxysmal Nocturnal Hemoglobinuria (PNH), an acquired genetic defect causes a deficiency in glycophosphatidylinositol on the membrane surface of red blood cells. This in turn leads to the immune system destroying its own red blood cells. So much so that after overnight concentration, urine has red color to it from the hemoglobin that has leaked from shredded cells – a tell-tall sign of Paroxysmal Nocturnal Hemoglobinuria and the orgin of the name. Thirty years old is the median age for diagnosis, and 80% of those diagnosed suffer organ failure or death within 3 years (typically from thrombosis or renal failure). Historically, palliative therapies like anticoagulants, blood and marrow transfusions, and supplements, have been the only form of treatment. More recently a monoclonal antibody called eculizumab (Soliris®) was approved to treat PNH has been shown to increase the quality of life for patients substantially relative to aforementioned treatments (it does not cure the disease). It is currently the most expensive drug in the world at $500,000 per year for ongoing treatment in the US, and more in other countries. The treatment is shrouded in controversy as the sponsor has held out or refused to supply the drug in some instances where there are subsidy disputes with government bodies in various countries demanding more justification for the record setting price. Clearly, this obviates the immediate and dire need for new drugs targeting PNH.
Bachem stands to make process improvements that can impact the cost of goods in such a way that it totally alters prescription habits – benefiting both courageous patients and their deserving sponsors.
MEET BACHEM: JULIE MARLEY
Site Manager at Bachem UK
How long have you been with Bachem? Where did you work before Bachem?
I have worked at Bachem for 17 years and was previously at Peninsula Laboratories Europe when it was acquired by Bachem.
Briefly, what do you do at Bachem?
I am responsible for managing all aspects of the BUK site.
What is your academic background/degrees or training?
I studied Chemistry at Liverpool, UK and subsequently obtained an MSc and PhD from part-time study whilst at Peninsula.
What do you like to do outside of work (interests, hobbies)?
I enjoy eating out with my family and friends enjoying the varied cuisines available in my local city and visiting the cinema.
What makes a perfect day for you?
When the combined contributions of all of the Bachem UK team result in the successful completion of an important project and exceed customer expectations.
What do you like most about your job?
I enjoy the range of responsibilities and the diverse challenges my role brings.
Have you had any particular expectation when you came to Bachem and have these been fulfilled?
I expected Bachem to be a highly professional organisation at the forefront of technological innovation and my expectations have been more than met.
What do you do for fun?
Gardening and cooking.
Thank you very much Julie.

PEPTIDE HIGHLIGHTS
Interesting news about peptides in basic research and pharmaceutical development:
Skin cells ‘crawl’ together to heal wounds treated with unique hydrogel layer-University of Toronto
New discovery may improve recovery after stroke-University of Gothenburg
Drugs from nature: big effects of multiple compounds in small amounts-Goethe University
The needle in the haystack– Technical University of Munich
LITERATURE CITATIONS
Bachem peptides and biochemicals are widely cited in research publications. Congratulations to all our customers with recent publications!
G. Calo, F. Sabbione, D. Vota, D. Paparini, R. Ramhorst, A. Trevani, C. Perez Leiros
Trophoblast cells inhibit neutrophil extracellular trap formation and enhance apoptosis through vasoactive intestinal peptide-mediated pathways
Hum. Reprod. 32 (1), 55-64 (2017)
C. Crewe, C. Schafer, I. Lee, M. Kinter, L. I. Szweda
Regulation of Pyruvate Dehydrogenase Kinase 4 in the Heart Through Degradation by the Lon Protease in Response to Mitochondrial Substrate Availability
J. Biol. Chem. 292(1), 305-312 (2017)
D. H. Nam, C. Rodriguez, A. G. Remacle, A. Y. Strongin and X. Ge
Active-site MMP-selective antibody inhibitors discovered from convex paratope synthetic libraries
Proc. Natl. Acad. Sci. U S A 113 (52), 14970-14975 (2016)
A. K. Saenger, O. Rodriguez-Fraga, R. Ler, J. Ordonez-Llanos, A. S. Jaffe, J. P. Goetze, F. S. Apple
Specificity of B-Type Natriuretic Peptide Assays: Cross-Reactivity with Different BNP, NT-proBNP, and proBNP Peptides
Clin. Chem. 63 (1), 351-358 (2017)
D. T. Tran, V. J. Cavett, V. Q. Dang, H. L. Torres and B. M. Paegel
Evolution of a mass spectrometry-grade protease with PTM-directed specificity
