DEFENSINS

Defensins are short cationic host-defense peptides and belong to the diverse class of antimicrobial peptides. Defensins are ribosomally synthesized and contain 6-8 conserved cysteine residues. They are found in vertebrates, invertebrates, plants, and in fungal species. More than 300 defensins have been identified so far.
The vertebrate defensins contain three disulfide bonds. They are categorized into three subfamilies, α-, β-, and θ-defensins on the basis of their size and disulfide bond pattern. α-Defensins comprise 29-35 amino acids and exhibit CysI-CysVI, CysII-CysIV, CysIII-CysV linkages whereas β-defensins have approximately 45 amino acids and display CysI-CysV, CysII-CysIV, CysIII-CysVI disulfide-bond patterns (Fig 1).
Figure 1 Disulfide Pattern of α- and β-Defensins
The characteristic disulfide patterns of α-defensins and β-defensins are illustrated by the peptides HNP-1 (human neutrophil peptide-1) as an example of α-defensins and HBD2 (human β-defensin 2) as an example of β-defensins.
Human α-Defensin HNP-1
Disulfide Pattern CysI-CysVI, CysII-CysIV, CysIII-CysV

Human β-Defensin 2 HBD2
Disulfide Pattern CysI-CysV, CysII-CysIV, CysIII-CysVI

θ-defensins are cyclic peptides of 18 amino acids. They are derived from two nonapeptide precursors, which are fused in a head-to-tail fashion. θ-defensins are characterized by three disulfide bonds in a CysI–CysVI, CysII–CysV, CysIII–CysIV arrangement (Fig 2). They are named like this because of their structural similarity to the Greek letter theta. The first θ-defensin was discovered in rhesus macaque (Macacca mulata) leukocytes and later on found in other non-human primates.
Figure 2 Rhesus macaque θ-Defensin
Disulfide pattern CysI–CysVI, CysII–CysV, CysIII–CysIV
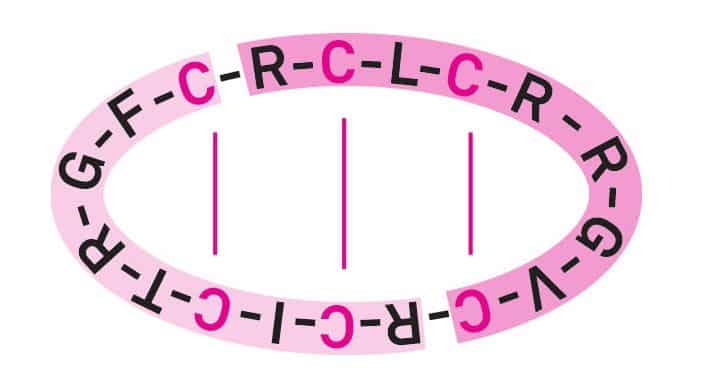
Adapted from A.C.Conibear, D.J.Craik, The chemistry and biology of theta defensins. Angew. Chem. Int. Ed. Engl. 2014, 53, 10612-10623.
Human Defensins
In humans, there are six α-defensins designated as DEFA 1-6. DEFA 1-4 are also known as HNP-1-4, DEFA 5 and 6 as human defensin HD5 and HD6.
HNP-1-4 are synthesized in promyelocytes, neutrophil precursor cells in the bone marrow, and stored in primary neutrophil granules as mature peptides whereas HD5 and HD6 are produced by Paneth cells of the small intestine but are also found in the reproductive tract.
Six human β-defensins (HBD1-6) have been characterized. A genomic approach, however, identified an additional 28 human β-defensin genes.
HBD1-3 are expressed ubiquitously by most epithelial surfaces but are also found in monocytes, macrophages, dendritic cells, and keratinocytes. The expression of HBD4 appears to be more restricted to the testes and gastric antrum. HBD5–6 are found in the epididymis.
Humans do not express θ-defensins. The human θ-defensin genes are pseudogenes. As the transcripts contain a premature stop codon the formation of θ-defensin peptides is prevented.
Mechanisms of Action
Defensins exhibit potent bactericidal activities against Gram-negative and Gram-positive bacteria. Besides membrane disruption by pore formation, defensins can interact with additional targets to induce bacterial death. Several mechanisms have been described. They may inhibit cell wall synthesis by binding to lipid II, an essential component of bacterial cell walls, interact with DNA and thereby blocking gene expression or capture bacteria by nanonet formation.
In addition to their antimicrobial effects, they show antiviral activities against a number of human viral pathogens such influenza A virus (IAV), human papillomavirus (HPV), herpes simplex virus type 1 (HSV-1), respiratory syncytial virus (RSV), and human immunodeficiency virus type 1 (HIV-1). Mechanisms like blocking receptor binding or membrane fusion, envelope disruption, and extracellular aggregation have been reported.
Apart from their effects on bacterial and viral pathogens, they also exhibit immunomodulatory effects including the stimulation of proinflammatory responses such as induction of proinflammatory cytokine production by macrophages and mast cells. They can also suppress inflammation through the inhibition of proinflammatory gene expression and cytokine production in response to bacterial infection. Apart from their immunomodulatory role, they are involved in the regulation of cell death pathways and cell survival.
Defensin Gene Polymorphism and Biomarkers
Single nucleotide polymorphisms and copy number variations in the defensin genes have been associated with susceptibility to various disorders. For instance, there is a significant association between the higher genomic copy numbers for the β-defensin genes and the relative risk of developing psoriasis. On the other hand, a lower HBD2 gene copy number in the β-defensin locus predisposes to colonic Crohn’s disease.
Single nucleotide polymorphisms of the HBD1 gene were connected with the pathogenesis of inflammatory bowel diseases and chronic gastritis, as well as oral diseases.
Recently, an α-defensin (HD5) gene single nucleotide polymorphism was shown to modulate the gut microbiota and thereby alter the risk of acute graft-versus-host disease. Polymorphism of defensin genes may also play a role in the susceptibility to infectious agents like HIV-1. It was demonstrated that HBD1 polymorphisms were significantly associated with decreased risk of HIV-1 infection acquisition and HIV-1 mother-to-child transmission.
As aberrant expression of defensins is present in various disease states, defensins may also serve as biomarkers. For instance, β-defensin 2 is of interest as a biomarker for psoriasis disease severity and potentially also as a post-treatment pharmacodynamic biomarker.
α-Defensins have been reported to serve as a biomarker for the diagnosis of periprosthetic joint infection (PJI), a serious complication after arthroplasty, and elevated levels of HD5 have been reported in Crohn’s disease.
Conclusions
Defensins are host defense peptides and play an important role in innate immunity and in the modulation of adaptive immune responses. Aberrant expression is linked to various disease states and therefore defensins represent potential biomarkers and targets for therapeutic intervention.
Bachem offers a range of readily available antimicrobial and antiviral peptides including defensins and can also support your research with an excellent custom synthesis service.
| Product Number | Product Name |
|---|---|
| 4025473 | Defensin HNP-1 (human) |
| 4025474 | Defensin HNP-2 (human) |
| 4025495 | Defensin HNP-3 (human) |
| 4102158 | Defensin 5 (human) |
| 4059148 | α-Defensin 6 |
| 4038285 | rec β-Defensin 1 (human) |
| 4038287 | rec β-Defensin 2 (human) |
| 4034693 | β-Defensin 2 (human) |
References
A.Rashidi, A.Herman, A.L.C.Gomes, J.U.Peled, R.R.Jenq, D.G.Brereton et al, An alpha-defensin gene single nucleotide polymorphism modulates the gut microbiota and may alter the risk of acute graft-versus-host disease. British Journal of Haematology 2020.
L.Zupin, V.Polesello, L.Segat, A.J.Kamada, L.Kuhn, S.Crovella, DEFB1 polymorphisms and HIV-1 mother-to-child transmission in Zambian population. The Journal of Maternal-Fetal & Neonatal Medicine 2019, 32, 2805-2811.
S.V.Prasad, K.Fiedoruk, T.Daniluk, E.Piktel, R.Bucki, Expression and function of host defense peptides at inflammation sites. International Journal of Molecular Sciences 2019, 21, 104.
S.Fruitwala, D.W.El-Naccache, T.L.Chang, Multifaceted immune functions of human defensins and underlying mechanisms. Seminars in Cell & Developmental Biology 2019, 88, 163-172.
M.Amerikova, I.Pencheva El-Tibi, V.Maslarska, S.Bozhanov, K.Tachkov, Antimicrobial activity, mechanism of action, and methods for stabilisation of defensins as new therapeutic agents. Biotechnology & Biotechnological Equipment 2019, 33, 671-682.
Q.Wu, J.Patocka, K.Kuca, Insect antimicrobial peptides, a mini review. Toxins (Basel) 2018, 10.
S.Sankaran-Walters, R.Hart, C.Dills, Guardians of the Gut: Enteric defensins. Frontiers in Microbiology 2017, 8, 647-647.
M.Pachón-Ibáñez, Y.Smani, J.Pachón, J.Sanchez-Cespedes, Perspectives for clinical use of engineered human host defense antimicrobial peptides. FEMS Microbiology Reviews 2017, 41, 323-342.
P.Chairatana, E.M.Nolan, Defensins, lectins, mucins, and secretory immunoglobulin A: microbe-binding biomolecules that contribute to mucosal immunity in the human gut. Critical Reviews in Biochemistry and Molecular Biology 2017, 52, 45-56.
F.Findlay, L.Proudfoot, C.Stevens, P.G.Barlow, Cationic host defense peptides; novel antimicrobial therapeutics against Category A pathogens and emerging infections. Pathogens and Global Health 2016, 110, 137-147.
A.C.Conibear, D.J.Craik, The chemistry and biology of theta defensins. Angewandte Chemie (International ed. in English) 2014, 53, 10612-10623.
A.L.Cole, A.M.Cole, The role of cationic polypeptides in modulating HIV-1 infection of the cervicovaginal mucosa. Antibiotics (Basel) 2014, 3, 677-693.
DEFENSINS IN DEVELOPMENT AS THERAPEUTICS
Defensins are endogenous antimicrobial peptides, which are known as innate defense molecules. The activity of defensins is focused on bacteria, fungi, and both enveloped and non-enveloped viruses. Their antiviral activities have been studied on many viruses including human immunodeficiency virus, human papillomavirus, herpes simplex virus, respiratory syncytial virus and several others (1). Some defensin-based therapeutics are in preclinical and clinical development as shown in Table 2.
| Product Name | Companies Involved | Highest Phase | Condition/Application |
|---|---|---|---|
| Brilacidin | Innovation Pharmaceuticals; Alfasigma | II | Skin and Skin Structure Infections (SSSI) Caused by Bacteria; Oral Mucositis; Proctitis |
| Human α-defensin 5 | Defensin Therapeutics ApS | Preclinical | Asthma; Inflammatory Bowel Disease; Metabolic Syndrome |
| Human β-defensin 2 | Defensin Therapeutics ApS; Dr. Falk Pharma | Preclinical | Asthma; Metabolic Syndrome; Crohn's Disease |
Phase II Candidate
Brilacidin, a defensin mimetic compound, is being developed by Innovation Pharmaceuticals and Alfasigma for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by Staphylococcus aureus, including Methicillin-resistant Staphylococcus aureus (MRSA) and Methicillin-sensitive Staphylococcus aureus (MSSA). This product candidate is also under development for oral mucositis and proctitis. Brilacidin is a bacterial cell membrane disrupter that selectively targets bacteria. In 2019, Innovation Pharmaceuticals announced that the company is planning to advance brilacidin in a Phase III oral mucositis program (1). There is also recent interest in evaluating brilacidin as a potential novel coronavirus (COVID-19) treatment. As a defensin-mimetic, brilacidin might bolster the body’s natural immune response to fight COVID-19 (2).
Preclinical Candidates
Defensin Therapeutics is developing human α-Defensin 5 (HD5) for the treatment of asthma, inflammatory bowel disease, and metabolic syndrome. The drug candidate is in preclinical development (1).
Defensin Therapeutics is also developing human β-Defensin 2 (HBD2). This drug candidate, a recombinant peptide, acts by targeting tumor necrosis factor-α (TNF-α). HBD2 binds to TNF-α and blocks its cytokine effects, thereby reducing TNF-induced inflammation. In 2016, Dr. Falk Pharma entered into a license agreement with Defensin Therapeutics that provides Dr. Falk Pharma with the rights to develop, manufacture, and market HBD2 in Europe and Australia (1).
Conclusion
Defensin-based therapeutics are attractive due to their favorable anti-bacterial, anti-viral, and anti-fungal characteristics. To support researchers and organizations who are studying and developing defensins, Bachem offers defensin peptides as research products. In addition, Bachem offers a comprehensive custom peptide synthesis service, and the production of New Chemical Entities.
References
1) M.S.Park et al. Toward the application of human defensins as antivirals. Biomol. Ther. (Seoul), 26(3) 242-254 (2018)
2) GlobalData (2020)
3) Scientific rationale for brilacidin as a potential novel coronavirus (COVID-19) treatment, Innovation Pharmaceuticals (2020)
MEET BACHEM: NI HUSI
What is your official job title at Bachem?
SAP Specialist Supply Chain Management.
How long have you been with Bachem? Where did you work before Bachem?
I have been with Bachem for four years and I worked for several pharmaceutical companies before Bachem.
Briefly, what do you do at Bachem?
I am responsible for two SAP modules to handle all the incidents and change requests related to these modules.
What is your academic background/degrees or training?
I have a Bachelor degree in public relations and English. I also have a Project Management Professional (PMP) certificate and SAP Material Management (MM) certificate.
What do you like to do outside of work?
I am fond of skiing, swimming, and hiking. I love to spend time in nature.
What makes a perfect day for you?
A day that starts with a smile.
What do you like most about your job?
Together with experts from each business area, finding the best solution in the SAP system to meet the business requirements
Have you had any particular expectation when you came to Bachem and have these been fulfilled?
I came to Bachem with a lot of years working in big companies, feeling that I was just one of the thousands of employees. Here at Bachem, it is more like a family and every family member’s contribution matters.
What do you do for fun?
Enjoy a wonderful evening with my girlfriends and skiing with my kids.
Would you like to communicate any key message to the reader?
I am very proud of being a member of the Bachem family.
Thank you very much Ni.

Peptide highlights
Interesting news about peptides in basic research and pharmaceutical development:
Foot-and-mouth-disease virus could help target the deadliest cancer-Queen Mary University of London
Alzheimer’s disease protein aggregation prevented by cell-penetrating peptides-Genetic Engineering & Biotechnology News
D6PV peptide lowers triglycerides in mouse bloodstream, shows study-Drug Target Review
We’re racing time’: biotech companies rush to complete coronavirus vaccine-WBUR
LITERATURE CITATIONS
Bachem peptides and biochemicals are widely cited in research publications. Congratulations to all our customers with recent publications!
A.Aidoukovitch et al.
Antimicrobial peptide LL-37 and its pro-form, hCAP18, in desquamated epithelial cells of human whole saliva.
European Journal of Oral Sciences128, 1-6 (2020)
L.Hu et al.
Threonine phosphorylation fine-tunes the regulatory activity of histone-like nucleoid structuring protein in Salmonella transcription.
Frontiers in Microbiology 10, (2019)
L.A.Stevens et al.
ADP-ribosylation of human defensin HNP-1 results in the replacement of the modified arginine with the noncoded amino acid ornithine.
Proceedings of the National Academy of Sciences 106, 19796 (2009)
S.Uchiyama et al.
Dual actions of group B Streptococcus capsular sialic acid provide resistance to platelet-mediated antimicrobial killing.
Proceedings of the National Academy of Sciences 116, 7465 (2019)
