Cysteine Derivatived Offered by Bachem
Cystine disulfide bridges help to stabilize the biologically active conformation of peptides and proteins. They are generated by incorporation of cysteine residues followed by oxidation of the thiol functions yielding disulfides („folding“). For the chemical synthesis of peptides, a range of protecting groups has been developed for blocking these sensitive moieties which may be removed either directly before or during oxidative folding.
When synthesizing peptides containing two or more disulfide bonds, S-protection may have to be varied to allow consecutive bridge formation for obtaining an unambiguous structure.
Synthesis of Cys-containing Peptides and Analogs
Peptides containing disulfide bridges
Albeit cysteine rarely occurs in bioactive peptides, its lateral thiol group is of utmost importance for stabilizing the tertiary structure due to its participation in disulfide bonds. Such links can be formed intra- and intermolecularly by oxidation. Protection of this very reactive moiety during the synthesis of peptides is mandatory. Hence the development of specifically cleavable thiol protecting groups has always been an issue in peptide chemistry.
Selective deblocking of the thiol moiety allows disulfide bond formation at various stages of the peptide assembly as well as the consecutive formation of two or more disulfide bridges 1D. Andreu et al. , Peptide Synthesis Protocols, p. 91, M.W. Pennington and B.M. Fields, eds., Humana Press, Totowa NJ (1994)2L. Moroder et al., Biopolymers 40, 207 (1996)3I. Annis et al., Methods Enzymol. 289, 198 (1997)4L. Chen et al., Current Protocols in Protein Science, Unit 18.6 (2001)5L. Moroder et al., Pept. Sci. 80, 85 (2004). In solution synthesis, the strategy has to be adapted to the presence of Cys (or Met) in the growing peptide, as sulfur-containing amino acids “poison” hydrogenation catalysts.
In solid-phase peptide synthesis (SPPS), thiol protection has to be compatible with the chosen strategy: trityl has become the most frequently used S-protection in Fmoc/ tBu-SPPS, whereas Boc/Bzl-SPPS requires S-protecting groups withstanding repeated contact with TFA. Mbzl or Mob are split off during the final HF cleavage. Acm, which is removed in a separate step, is compatible with both strategies.
Cysteine Derivatives
Bachem offers a broad selection of protected cysteine derivatives for most synthetic needs. Additionally, you will find a choice of derivatives of cystine, homocysteine, penicillamine, and thioproline in this brochure. Moreover, we offer a range of protected mercapto carboxylic acids, useful building blocks for synthesizing analogs of cystinecontaining peptides.
Furthermore, removal of S-protection and oxidation yielding the disulfide bridge may be performed either consecutively or concomitantly, by oxidative deprotection. If two or more disulfide bonds have to be formed either pairwise orthogonal protection is used allowing selective, consecutive disulfide bridge formations or a single Cys protecting group is chosen. Then all the sulfhydryls are liberated simultaneously followed by bridging in buffered solutions containing redox systems such as oxidized and reduced glutathione; the term “oxidative folding” has been coined for this approach.
Clearly, oxidative folding is the more convenient approach, but bioactive peptides are often derived from precursors, which are oxidized before being cleaved to generate the active circulating form. In such cases, the correct bridging may only be obtained by consecutive disulfide bond formation.
Disulfide bridges stabilize the tertiary structures of proteins
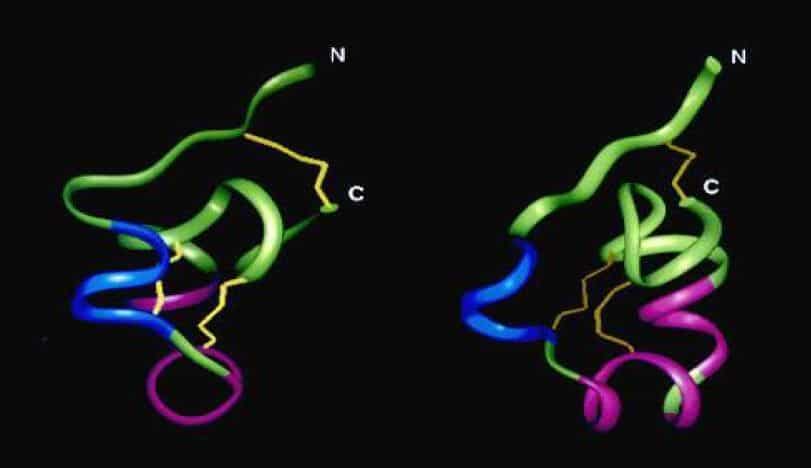
ShK toxin, a 35-residue peptide toxin isolated from the sea anemone Stichodactyla helianthus, contains three intramolecular disulfide bonds to stabilize the compactly folded biologically active conformation.
This also applies for analogs and unnatural folding patterns.6B. Hargittai and G. Barany, J. Pept. Res. 54, 468 (1999) Peptides containing a single intermolecular disulfide bond, i.e. dimers, can be obtained using any Cys protecting group compatible with the chosen combination of Nα/sidechain protecting groups. A different strategy is required when synthesizing heterodimers. SH-labile thiol protecting groups as NPys (often introduced post-synthetically)7J. Ottl and L. Moroder, Tetrahedron Lett. 40, 1487 (1999) allow the selective bridging of different peptide chains8R. Matsueda et al., Chem. Lett. 737 (1981); M.S. Bernatowicz et al., Int. J. Pept. Protein Res. 28, 107 (1986).
For modifying disulfide bridges, one Cys residue or both can be replaced by other β-mercapto amino acids as penicillamine (Pen) or Cys homologs as homocysteine (Hcy). A considerable number of analogs of bioactive peptides, e.g. vasopressin and oxytocin antagonists, has been obtained by substituting an N-terminal Cys by β mercaptopropionic acid or β-mercapto-β,β-cyclopentamethylenepropionic acid. Cysteamine can replace C-terminal Cys. Thioether bridges can be obtained by exploiting the high nucleophilicity of the sulfhydryl group, which smoothly reacts with lateral haloacetyl groups in the presence of a base.9F.M. Brunel and P.E. Dawson, Chem. Commun. 2552 (2005) Peptides containing Cys and dehydroalanine yield lanthionine peptides upon cyclization.
Peptides containing unbridged cysteine
Besides its role in disulfide bridge formation, the sulfhydryl group of cysteine has further important biological functions, e.g. in the active site of SH-proteases. Moreover, it can be nitrosylated, palmitoylated, or prenylated posttranslationally. The peptide chemist can benefit as well from this highly reactive moiety allowing a broad spectrum of selective modifications.
Peptides containing an exposed single free sulfhydryl moiety, usually a C- or N-terminal cysteine, are used to prepare peptideprotein conjugates. The link is generated by reacting the thiol with maleimide bound to the carrier. Please ask about our custom synthesis services for conjugation to KLH, BSA or thyroglobulin carrier protein.
Peptides containing several free thiol moieties can form complexes with metal ions such as Zn(II) (“zinc finger peptides”), Cu(I) and Ag(I). They are readily obtained by SPPS using a single SH protecting group, but they have to be protected from (random) oxidation.10F. Cornille et al., Int. J. Pept. Protein Res. 36, 551 (1990)
Activation and coupling of Cys derivatives
Cys derivatives are notorious for basecatalyzed racemization during activation and coupling.11Y. Han et al., J. Org. Chem. 62, 4307 (1997)12Y.M. Angell et al., J. Pept. Res. 60, 292 (2002)
Considerable amounts of D-Cys epimer are obtained when coupling Cys(Trt) derivatives in the presence of bases. Cys(Acm) derivatives show a lower tendency to racemize, they tolerate weak bases as collidine. Attempted syntheses of peptides containing several disulfide bridges following standard Fmoc protocols may have failed for this reason. The extent of this side-reaction can be reduced by using weak bases as collidine in combination with uronium/aminium or phosphoniumreagents or, more effectively, by coupling in the absence of bases, e.g. with carbodiimides and HOBt (or HOAt).
Racemization is further impeded by using less polar solvents for the coupling. Further side reactions of cysteine during peptide synthesis have been described in the literature, for a review see e.g.13L. Moroder et al., Methods of Organic Chemistry (Houben-Weyl), Vol. E 22a, Synthesis of Peptides and Peptidomimetics, p. 384, M. Goodman, A. Felix, L. Moroder and C. Toniolo, eds., Georg Thieme Verlag, Stuttgart (2002)
Usually, the extent of by product formation from Cys depends on the nature of the thiol protecting group. This aspect should not be ignored when developing a synthetic strategy for peptides containing several Cys residues.
The role of Cys in Native Chemical Ligation
The cysteine sulfhydryl group plays a crucial role in the synthesis of large peptides by Native Chemical Ligation (NCL)14T.M. Hackeng et al., Proc. Natl. Acad. Sci. USA 96, 10068 (1999)15D. Macmillan, Angew. Chem. Int. Ed. 45, 7668 (2006).
The key step of NCL consists of the reaction of a peptide thioester with a peptide containing an N-terminal Cys. When assembling small proteins from three or more peptide fragments, the middle fragments have to be N-terminally protected to avoid oligomerization or an intramolecular reaction of the thioesters.
Thz (thiazolidine-4-carboxylic acid) can be attached to such fragments as an equivalent for N-terminal Cys, as the thioaminal may be cleaved under mild conditions liberating the mercapto and amino moieties concomitantly16M. Villain et al., Peptides, the Wave of the Future, Proceedings 2nd International and17th American Peptide Symposium, San Diego 2001, p. 107, M. Lebl and R. Houghten, eds., Kluwer Scientific Publisher, Dordrecht (2007)17D. Bang and S.B.H. Kent, Angew. Chem. Int. Ed. Engl. 43, 2534 (2004).
Thiol Protection During Solid-Phase Synthesis
Thiol protecting groups
In Fmoc-based SPPS, Trt18G. Amiard and L. Velluz, Bull. Soc. Chim. Fr. 698 (1956); L. Velluz et al., Bull. Soc. Chim. Fr. 1464 (1956); L. Zervas et al., J. Am. Chem. Soc. 78, 1359 (1956) is the preferred protecting group for the SH-moiety. In the presence of scavengers such as EDT Trt is smoothly removed with TFA. Additionally, Mob19S. Akabori et al., Bull. Chem. Soc. Jpn. 37, 443 (1964), the very acid-labile Mmt20K. Barlos et al., Int. J. Pept. Protein Res. 47, 148 (1996) or the orthogonal Acm21D.F. Veber et al., Tetrahedron Lett. 3057 (1968); D.F. Veber et al., J. Am. Chem. Soc. 94, 5456 (1972) are frequently chosen for the synthesis of more complex Cys-containing peptides.
If linked to the resin by an ester bond, C-terminal cysteine is prone to base-induced β-elimination followed by addition of piperidine during the subsequent Fmoc cleavage steps. CTerminal Cys(Trt) should be preferred over Cys(Acm) in order to reduce the extent of this side reaction22J. Lukszo et al., Lett. Pept. Sci. 3, 157 (1996), especially in case of an adjacent Ser or Thr. Fmoc-Asp(OtBu)-OH should not be coupled to Cys(Acm), as the Asp(OtBu)-Cys(Acm) motif readily forms aspartimide in the presence of bases.
The motif Asp(OtBu) Cys(Trt) turned out to be far less sensitive towards the repetitive piperidine treatments during Fmoc-based SPPS23M. Mergler et al., J. Pept. Sci. 9, 518 (2003). Such limitations have to be kept in mind when developing strategies for consecutive disulfide bridge formation. Mob and Mbzl24B.W. Erickson and R.B. Merrifield, J. Am. Chem. Soc. 95, 3750 (1973) are the most commonly used protecting groups for the sulfhydryl function in Boc-based SPPS. Mbzl is more stable towards the repetitive acidolysis of the Nα-deprotection step. Hence it should be chosen when synthesizing long peptides. Both groups are removed during the final cleavage with HF. Orthogonal thiol protection is attained by selecting Acm, StBu25U. Weber and P. Hartter, Hoppe-Seyler’s Z.
Physiol. Chem. 351, 1384 (1970), NPys, or base-labile Fm26M. Ruiz-Gayo et al., J. Chem. Soc. Chem. Commun. 1501 (1986).
Formation of Disulfide Bridges by Air Oxidation The peptide is dissolved at a concentration of 10-3 to 10-4 M in dilute acetic acid or 0.05 M ammonium acetate; the pH is adjusted and kept between 7.5 and 8 whilst stirring the solution at room temperature in the presence of atmospheric oxygen.
Additives such as 1-10% DMSO or 3% H2O2 can be used to accelerate the reaction (H2O2 must not be used if the peptide contains Met). The progress of the reaction is followed by HPLC.
The pH has to be checked regularly and readjusted if required. After complete conversion, the solution is acidified with acetic acid. It can be applied directly to preparative HPLC.
Peptides containing a single disulfide bridge
When applying Fmoc-Cys(Trt)-OH for incorporating both cysteines, cleavage with TFA/scavengers yields the crude deprotected linear peptide, which may be cyclized directly by atmospheric oxygen or other mild oxidants (e.g. potassium hexacyanoferrate (III)). The presence of scavengers during TFA cleavage is mandatory. Silanes as Et3SiH are most efficient in removing the trityl cations as they reduce them to inert triphenylmethane. Usually, the oxidation is performed in very dilute solution to favor intramolecular reaction.
| Protecting group | Cleavage conditions | Remarks |
|---|---|---|
| Trt | TFA/ scavengers | standard (Fmoc) |
| Mmt | 1%TFA/DCM/EDT or silanes | selective deprotection of Cys |
| Acm | Ag(I), Hg(II) | orthogonal to Fmoc and Boc |
| Dpm1) | TFA/scavengers | Fmoc |
| Mob | HF | standard (Boc) |
| Mbzl | HF | standard (Boc) |
| Fm | secondary amine | orthogonal, withstands HF |
| NPys | thiolysis / reduction / S-S interchange | orthogonal, withstands HF |
| StBu | reduction | orthogonal, withstands HF |
1) Use of Dpm in place of Trt [42] also reduces oxidation of Cys to cysteic acid.
Fmoc-Cys(Acm)-OH is chosen when conducting cleavage and cyclization consecutively, with the option of purifying the crude S-protected peptide in between. On the other hand, the very acid-labile, selectively cleavable Mmt allows the oxidation of otherwise protected fragments. It can also be removed on-resin (e.g. on Wang resin).
In Boc/Bzl-based SPPS followed by HFcleavage, use of Mob or Mbzl (the latter being more stable towards the repetitive TFA-treatments during the synthesis) yields the free peptide. Use of Acm yields an Acm-protected crude peptide, which may be oxidized with iodine. For a comparison of various oxidation methods (in solution and on-resin) see27J. Eichler and R.A. Houghten, Protein Pept. Lett. 4, 157 (1997).
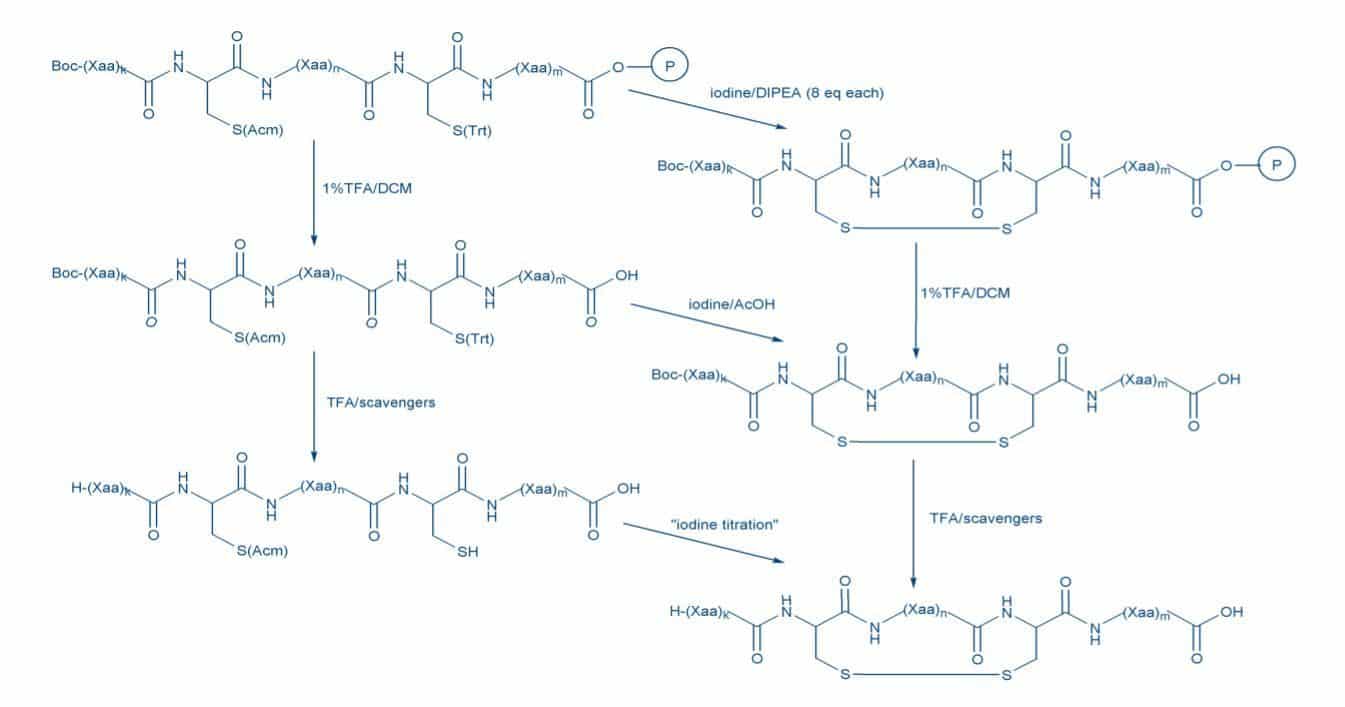
Strategies for the Fmoc-SPPS of a peptide containing a single disulfide bridge employing a highly acid-labile resin, e.g. SASRIN or 2-chlorotrityl resin.
Peptides containing two disulfide bridges
Mispairings, which may occur when oxidizing the four free sulfhydryl groups concomitantly, can be excluded by consecutive formation of the disulfide bridges. The adequate combination of S-protecting groups allows specific cleavage and the use of optimal cyclization conditions. The order, in which the disulfide bonds are formed, may decide the outcome of the synthesis. In most cases, the “smaller cycle” is generated first.
| Cysteine protecting groups | Cleavage of cysteine protecting groups | Cyclization conditions |
|---|---|---|
| Cys(Trt) + Cys(Trt) | 95% aq. TFA+5% EDT | standard, separate steps, liberation of SH followed by oxidation followed by oxidation (air, H2O2, iodine,…) |
| Cys(Acm) + Cys(Acm) | iodine in 80% AcOH1) | standard, oxidative cleavage, orthogonal to tBu/Wang |
| Cys(Trt) + Cys(Acm) | 95% aq. TFA+5% EDT; 1 eq. iodine in 80% AcOH (iodine titration) | separate steps, Acm is cleaved concomitantly with bridge formation by titration with iodine |
1) Precautions have to be taken in the presence of Tyr, His, Met and especially Trp. Trp can be protected by adding a large excess of Ac-Trp- OMe. Protected peptides are less susceptible to iodine-induced side-reactions.
Several combinations of protecting groups may be chosen. 28D. Andreu et al., Peptide Synthesis Protocols, p. 91, M.W. Pennington and B.M. Fields, eds., Humana Press, Totowa NJ (1994)29L. Moroder et al., Biopolymers 40, 207 (1996)30I. Annis et al., Methods Enzymol. 289, 198 (1997)31L. Chen et al., Current Protocols in Protein Science, Unit 18.6 (2001)32L. Moroder et al., Pept. Sci. 80, 85 (2004)33J. Eichler and R.A. Houghten, Protein Pept. Lett. 4, 157 (1997)We have described in our “Recommended Standard Procedure” protocol the most used combination Trt/ Acm depicted below:
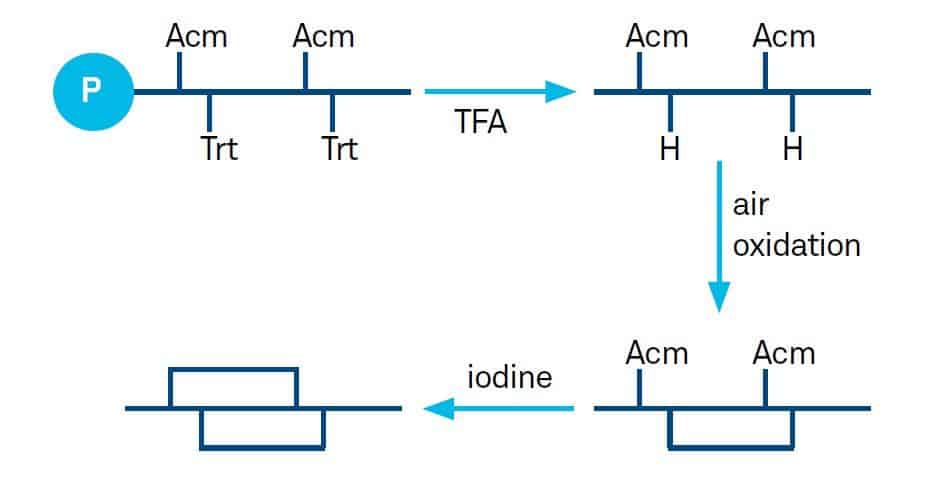
Formation of Disulfide Bridges from Bis-Acm Peptides The peptide is dissolved at a concentration of 10-3 to 10-4 M in 40% aqueous acetic acid. Iodine (25 to 50 fold excess) dissolved in 40% acetic acid (or in methanol) is added. The solution is stirred at room temperature, the progress of the reaction is monitored by analytical HPLC.
To stop the reaction the excess of iodine is destroyed with 1 M aqueous ascorbic acid (the solution is added slowly up to disappearance of the iodine color). The solution is diluted with water to lower the concentration of acetic acid to approximately 10% before applying it to a preparative HPLC column.
Peptides containing three disulfide bridges
The combination Acm/Mob/Trt has been successfully used for the preparation of several peptides (amongst them relaxin34E.E. Büllesbach and C. Schwabe, J. Biol. Chem. 266, 10754 (1991), defensins35J.P. Durieux and R. Nyfeler, Peptides 1994, Proceedings of the 23rd European Peptide Symposium, Braga 1994, p. 165, H.L.S. Maia, ed., ESCOM Publishers, Leiden (1995), sapecin36M. Mergler and R. Nyfeler, Proceedings of the 4th International Symposium on Innovation and Perspectives in SPPS, Edinburgh 1995, p.485, R. Epton, ed., Mayflower Scientific Ltd., Birmingham (1996)) containing three disulfide bridges employing Fmoc/tBu-SPPS and selective bridging. It should be kept in mind that the consecutive formation of the disulfide bridges is a long and tedious process.
If the peptide to be prepared corresponds to a naturally occurring sequence, not excised from a prepeptide and showing the natural bridging pattern, the simultaneous formation of disulfide bridges (random oxidation) can render very good results37J.P. Durieux and R. Nyfeler, Peptides, Chemistry, Structure and Biology, Proceedings 14th American Peptide Symposium, Columbus 1995, p.42, P.T.P. Kaumaya and R.S. Hodges, eds., Mayflower Scientific Ltd. (1996)38Z. Wu et al., J. Pept. Res. 64, 118 (2004). Both methods can be combined39S.E. Escher et al., J. Pept. Res. 54, 505 (1999).
Simultaneous formation of disulfide bridges
Using this approach a single kind of protecting group must be chosen for the protection of the sulfhydryl function. In Fmoc-based SPPS, Trt is the most commonly used thiol protection cleaved during the final TFA deprotection; Acm or Mbzl have been used in Boc SPPS. The crude peptide can be prepurified or subjected directly to oxidation. The Acm group is stable to acidolysis. Hence the SH moieties of the peptide obtained after the final TFA (or HF) deprotection still are blocked, which facilitates the purification.
| Pairing of Cys protecting groups | Cleavage of Cys protecting group | Cyclization conditions |
|---|---|---|
| Cycle 1: Cys(Trt) | TFA/H2O2/scavengers | Air oxidation |
| Cycle 2: Cys(Acm) | Concomitant cleavage and cyclization with iodine | |
| Cycle 3: Cys(Mob) | TFMSA/TFA/anisole+5%EDT) | Air or DMSOoxidation, or iodine titration |
The Acm group can then be cleaved by mercuric acetate followed by oxidative folding. The reaction is mediated by redox systems such as reduced and oxidized glutathione, cysteine/cystine or DTT/oxidized DTT. This approach has been used in the preparation of muscarinic toxin 1 (MTX1)40Y. Nishiuchi et al., J. Pept. Sci. 6, 84 (2000) a 66-resi-due peptide containing 5 disulfide bridges following a convergent Boc synthesis based on a combined solid phase-solution approach.
However, folding does not yield automatically the correct disulfide bridging. In certain cases, a product containing an unnatural folding pattern could be isolated predominantly at first.41S. Kubo et al., Biopolymers 38, 733 (1996)The fine-tuning of the cyclization conditions (reaction at 5°C in the presence of 2 M sodium sulfate) has resulted in the isolation of the correctly folded, bioactive molecule as the main product.
On-Resin Disulfide Bridge Formation The resin is left to swell in DCM/MeOH/H2O (60:25:4,v/v/v; 18 ml/g resin) for 0.5 to 1 hr before adding 8 eq I2 in DCM (12 ml/g resin) and 8 eq DIPEA Reaction time: 1 hr or less. Washes: DMF, aq. ascorbic acid/ DMF, aq. DMF, DMF, DCM or IPA.
On-resin cyclization
Peptides containing a single disulfide bridge can be obtained via SPPS by various synthetic strategies, which are summarized in the scheme on p. 6 (for SASRIN or other highly acid-sensitive resins). Either the (partially) protected peptide or the deprotected peptide may be oxidized in solution, but as to enhance intramolecular disulfide formation, the oxidation is performed in very dilute solution thus making work-up rather tedious.
Oxidation of a resin-bound peptide circumvents this problem. Quite a number of papers dealing with variations of this approach as employing different types of resin, sulfhydryl protecting groups and oxidants has been published,42R. Albert et al., Bioorg. Med. Chem. Lett. 8, 1207 (1998)though, to our knowledge, a systematic overview is still lacking.
Cleavage of Mob and Disulfide Bridge Formation The cleavage cocktail (TFMSA/TFA/anisole 1:8:1) is prepared and cooled in an ice bath. The cooled mixture is added to the peptide (3 mg/ml). The resulting solution is stirred at 0°C for 45 min.
The reaction solution is diluted 50-fold with ice-cold water and extracted 3 times with ether. DMSO (10% of total volume) is added to the aqueous phase. The cyclization is monitored by analytical HPLC. If complete conversion is achieved, the reaction solution is diluted with water and used directly for preparative HPLC purification.
The cyclization can also be performed by iodine titration: addition of a methanolic solution of iodine up to persistent yellow colour and destruction of the excess of iodine with ascorbic acid.
Cyclization yields depend on several factors, amongst them the peptide load (a low load should reduce intermolecular reaction and thus enhance cyclization, i.e. the pseudodilution effect), the size of the ring to be formed (probably best below 10 amino acids), and the solvent which should properly swell the peptide resin. Oxidations proceeding smoothly in solution may also work well if performed with the resin-bound peptide (for a noteworthy exception see)43M. Kakiuchi et al., Chem. Pharm. Bull. 44, 1107 (1996). The N-terminus should be protected.
When oxidizing peptides linked to highly acid-sensitive carriers (e.g. SASRIN, ClTrtresin) with iodine, the concomitantly formed HI should be neutralized. At Bachem, oxidative cyclization has been performed successfully on SASRIN. The conditions described here have been evaluated for [Cys1(Acm), Cys7(Trt)]-salmon calcitonin (1-10) and [Cys2(Acm or Trt), Cys7(Trt)]-α-CGRP (human) (1-14)44R. Kamber et al., Helv. Chim. Acta 63, 899 (1980).
The resin (load of fully protected peptide approximately 0.1 mmol/g or below) is left to swell in DCM/MeOH/H2O (60:25:4, approx. 18 ml/g resin) for at least 30 minutes. A solution of iodine (8 eq) in DCM (12 ml/g resin) is added rapidly, followed by DIPEA (max 8 eq) to neutralize the hydrogen iodide evolved during the reaction. Oxidation time should not exceed one hour, the rate has to be determined by taking samples and quenching them with ascorbic acid before cleavage and analysis. Eventually excess iodine is removed by filtering and washing the resin with DMF and DMF/ascorbic acid in aqueous buffer of pH7.
Nevertheless the resin may remain slightly yellow. As usual, all polar impurities have to be removed before cleaving with 1% TFA/DCM. The yield obtained may be lower than the cleavage yield of the linear fully protected peptide. Yields depend on the conditions of oxidation, especially on the solvent, thus preliminary experiments optimizing this parameter should be conducted if possible. Other synthetic strategies may be chosen, e.g. generation of sulfhydryl groups and oxidation as separate steps.
If S-t-butylthio protection is employed the peptide resin is first treated with tributylphosphine45R. Eritja et al., Tetrahedron 43, 2675 (1987)46L. Moroder et al., Hoppe-Seyler’s Z. Physiol. Chem. 362, 697 (1981) (or β-mercaptoethanol), thoroughly washed and then treated with an oxidant. The incorporation of Cys(Mmt) allows consecutive on-resin deprotection and cyclization during Fmoc-SPPS47K. Barlos et al., Int. J. Pept. Protein Res. 47, 148 (1996). Further modes of on-resin cyclization involving Cys, e.g. via thioether bond,48K.D. Roberts et al., Tetrahedron Lett. 39, 8357 (1998) have been described in the literature.
Subscribe to our newsletter
"*" indicates required fields
General References
J.M. Stewart and J.D. Young, eds.
Solid Phase Peptide Synthesis
Pierce Chemical Company, Rockford, Illinois (1984)
M.W. Pennington and B.M. Dunn, eds.
Peptide Synthesis Protocols
Humana Press, Totowa (1994)
P. Lloyd-Williams, F. Albericio, E.
Giralt Chemical Approaches to the Synthesis of Peptides and Proteins
CRC Press, Boca Raton (1997)
W.C. Chan and P.D. White, eds.
Fmoc Solid Phase Peptide Synthesis. A Practical Approach
Oxford University Press, Oxford (2000)
S.A. Kates and F. Albericio, eds.
Solid-Phase Synthesis, a Practical Guide
Marcel Dekker, Inc., New York, Basel (2000)
N. Sewald and H.D. Jakubke, eds.
Peptides: Chemistry and Biology
Wiley-VCH Verlag GmbH, Weinheim (2002)
M. Goodman, A. Felix, L. Moroder, C. Toniolo, eds.
Synthesis of Peptides and Peptidomimetics
(Methods of Organic Chemistry (Houben-Weyl), Vol. E 22a and E 22b)
Georg Thieme Verlag, Stuttgart (2002)
T.W. Greene and P. G.M. Wuts, eds.
Greene‘s Protective Groups in Organic Synthesis, 4th ed.
John Wiley & Sons, Inc., New York (2007)
Litterature References
- 1D. Andreu et al. , Peptide Synthesis Protocols, p. 91, M.W. Pennington and B.M. Fields, eds., Humana Press, Totowa NJ (1994)
- 2L. Moroder et al., Biopolymers 40, 207 (1996)
- 3I. Annis et al., Methods Enzymol. 289, 198 (1997)
- 4L. Chen et al., Current Protocols in Protein Science, Unit 18.6 (2001)
- 5L. Moroder et al., Pept. Sci. 80, 85 (2004)
- 6B. Hargittai and G. Barany, J. Pept. Res. 54, 468 (1999)
- 7J. Ottl and L. Moroder, Tetrahedron Lett. 40, 1487 (1999)
- 8R. Matsueda et al., Chem. Lett. 737 (1981); M.S. Bernatowicz et al., Int. J. Pept. Protein Res. 28, 107 (1986)
- 9F.M. Brunel and P.E. Dawson, Chem. Commun. 2552 (2005)
- 10F. Cornille et al., Int. J. Pept. Protein Res. 36, 551 (1990)
- 11Y. Han et al., J. Org. Chem. 62, 4307 (1997)
- 12Y.M. Angell et al., J. Pept. Res. 60, 292 (2002)
- 13L. Moroder et al., Methods of Organic Chemistry (Houben-Weyl), Vol. E 22a, Synthesis of Peptides and Peptidomimetics, p. 384, M. Goodman, A. Felix, L. Moroder and C. Toniolo, eds., Georg Thieme Verlag, Stuttgart (2002)
- 14T.M. Hackeng et al., Proc. Natl. Acad. Sci. USA 96, 10068 (1999)
- 15D. Macmillan, Angew. Chem. Int. Ed. 45, 7668 (2006)
- 16M. Villain et al., Peptides, the Wave of the Future, Proceedings 2nd International and17th American Peptide Symposium, San Diego 2001, p. 107, M. Lebl and R. Houghten, eds., Kluwer Scientific Publisher, Dordrecht (2007)
- 17D. Bang and S.B.H. Kent, Angew. Chem. Int. Ed. Engl. 43, 2534 (2004)
- 18G. Amiard and L. Velluz, Bull. Soc. Chim. Fr. 698 (1956); L. Velluz et al., Bull. Soc. Chim. Fr. 1464 (1956); L. Zervas et al., J. Am. Chem. Soc. 78, 1359 (1956)
- 19S. Akabori et al., Bull. Chem. Soc. Jpn. 37, 443 (1964)
- 20K. Barlos et al., Int. J. Pept. Protein Res. 47, 148 (1996)
- 21D.F. Veber et al., Tetrahedron Lett. 3057 (1968); D.F. Veber et al., J. Am. Chem. Soc. 94, 5456 (1972)
- 22J. Lukszo et al., Lett. Pept. Sci. 3, 157 (1996)
- 23M. Mergler et al., J. Pept. Sci. 9, 518 (2003)
- 24B.W. Erickson and R.B. Merrifield, J. Am. Chem. Soc. 95, 3750 (1973)
- 25U. Weber and P. Hartter, Hoppe-Seyler’s Z.
Physiol. Chem. 351, 1384 (1970) - 26M. Ruiz-Gayo et al., J. Chem. Soc. Chem. Commun. 1501 (1986)
- 27J. Eichler and R.A. Houghten, Protein Pept. Lett. 4, 157 (1997)
- 28D. Andreu et al., Peptide Synthesis Protocols, p. 91, M.W. Pennington and B.M. Fields, eds., Humana Press, Totowa NJ (1994)
- 29L. Moroder et al., Biopolymers 40, 207 (1996)
- 30I. Annis et al., Methods Enzymol. 289, 198 (1997)
- 31L. Chen et al., Current Protocols in Protein Science, Unit 18.6 (2001)
- 32L. Moroder et al., Pept. Sci. 80, 85 (2004)
- 33J. Eichler and R.A. Houghten, Protein Pept. Lett. 4, 157 (1997)
- 34E.E. Büllesbach and C. Schwabe, J. Biol. Chem. 266, 10754 (1991)
- 35J.P. Durieux and R. Nyfeler, Peptides 1994, Proceedings of the 23rd European Peptide Symposium, Braga 1994, p. 165, H.L.S. Maia, ed., ESCOM Publishers, Leiden (1995)
- 36M. Mergler and R. Nyfeler, Proceedings of the 4th International Symposium on Innovation and Perspectives in SPPS, Edinburgh 1995, p.485, R. Epton, ed., Mayflower Scientific Ltd., Birmingham (1996)
- 37J.P. Durieux and R. Nyfeler, Peptides, Chemistry, Structure and Biology, Proceedings 14th American Peptide Symposium, Columbus 1995, p.42, P.T.P. Kaumaya and R.S. Hodges, eds., Mayflower Scientific Ltd. (1996)
- 38Z. Wu et al., J. Pept. Res. 64, 118 (2004)
- 39S.E. Escher et al., J. Pept. Res. 54, 505 (1999)
- 40Y. Nishiuchi et al., J. Pept. Sci. 6, 84 (2000)
- 41S. Kubo et al., Biopolymers 38, 733 (1996)
- 42R. Albert et al., Bioorg. Med. Chem. Lett. 8, 1207 (1998)
- 43M. Kakiuchi et al., Chem. Pharm. Bull. 44, 1107 (1996)
- 44R. Kamber et al., Helv. Chim. Acta 63, 899 (1980)
- 45R. Eritja et al., Tetrahedron 43, 2675 (1987)
- 46L. Moroder et al., Hoppe-Seyler’s Z. Physiol. Chem. 362, 697 (1981)
- 47K. Barlos et al., Int. J. Pept. Protein Res. 47, 148 (1996)
- 48K.D. Roberts et al., Tetrahedron Lett. 39, 8357 (1998)